Transmissionputty
putty 时间:2021-01-25 阅读:()
PI–78,Rev4,08/2016RM-1479READBEFOREUSINGDBMPuttyDEMINERALIZEDBONEMATRIXDONATEDHUMANTISSUECAUTION:DEVICEISFORSINGLEPATIENTUSEONLY.
AsepticallyProcessed.
PassesUSPSterilityTests.
DBMPuttyIsNotTerminallySterilized.
DESCRIPTIONDBMPuttyisprocessedhumanbonethathasbeendemineralizedandcombinedwithsodiumhyaluronate,whichisanaturallyderivedmaterialnotofanimaloriginthatisbothbiocompatibleandbiodegradable.
Thecombinationofdemineralizedboneandsodiumhyaluronateresultsinaputty-likeortexturedconsistencyforeaseandflexibilityofuseduringsurgicalapplication.
OSTEOINDUCTIVEPOTENTIALDBMPuttyisosteoconductive,andhasbeenshowntohaveosteoinductivepotentialinanathymicmousemodel.
EverylotoffinalDBMPuttyproductistestedinanathymicmousemodelorinanalkalinephosphataseassay,whichhasbeenshowntohaveapositivecorrelationwiththeathymicmousemodel,toensuretheosteoinductivepotentialofthefinalproduct.
Standardtestingperformedinanathymicmouseoralkalinephosphataseassaymustprovepositiveforlotrelease.
Itisunknownhowtheosteoinductivepotential,measuredintheathymicmousemodelorthealkalinephosphataseassay,willcorrelatewithclinicalperformanceinhumansubjects.
INDICATIONSFORUSEDBMPuttyisintendedfortheaugmentationofdeficientmaxillaryandmandibularalveolarridgesandthetreatmentoforal/maxillofacialanddentalintraosseousdefectsincluding:-Ridgeaugmentation-Fillingofcysticdefect-Fillingofextractionsites-Fillingoflesionsofperiodontalorigin-Craniofacialaugmentation-FillingofdefectsofEndodonticorigin-Mandibularreconstruction-Repairoftraumaticdefectsofthealveolarridge,excludingmaxillaryandmandibularfracture-Fillingresectiondefectsinbenignbonetumors,benigncystsorotherosseousdefectsinthealveolarridgewallDBMPuttycanbeusedwithautograftorbonemarrowaspirate.
DBMPuttyisforsinglepatientuseonly.
CONTRAINDICATIONSDBMPuttyisNOTintendedtoprovidestructuralsupportoftheboneduringthehealingprocess.
DBMPuttyisalsocontraindicatedforincompletemaxillofacialskullgrowth.
ADVERSEEFFECTSPossibleadverseeffectsofusingDBMPuttyinclude,butarenotlimitedto:PotentiallossofcontourofmaxillofacialskullInfectionofsofttissueand/orbone(osteomyelitis)FeverDeformityoftheboneatthesiteIncompleteboneingrowth,delayedunionornon-unionHypercalcemiaortransienthypercalcemiaFractureofthenewlyformedboneDiseasetransmissionandundesirableimmuneresponseWithintheUnitedStates:AdverseoutcomesattributabletothetissuemustbepromptlyreportedtoMTF.
OutsideoftheUnitedStates:Adverseoutcomesattributabletothetissuemustbepromptlyreportedtoyourlocalrepresentative.
CAUTIONSDonotsterilize.
Donotfreeze.
DBMPuttymayextrudeintofacialsofttissue.
TraceamountsofGentamicinmaybepresent.
Tissueisexposedtoprocessingsolutionsthatmaycontaindetergentsandalcohol.
Traceamountsofprocessingsolutionmayremain.
Cautionshouldbeexercisedifthepatientisallergictoanyofthesesubstances.
NOTE:No-lactamantibioticsareusedduringtheprocessingoftissueinDBMPuttyproducts.
ExtensivemedicalscreeningprocedureshavebeenusedintheselectionofalltissuedonorsforMTF(pleaseseeDonorScreeningandTesting).
TransmissionofinfectiousdiseasessuchasHIVorhepatitis,aswellasatheoreticalriskoftheCreutzfeldt-Jakob(CJD)agent,mayoccurinspiteofcarefuldonorselectionandserologicaltesting.
Closedsuctionordrainageisrecommendedtopreventfluidaccumulationinthewound.
Cautionshouldbetakenforthefollowingcircumstances:SeverevascularorneurologicaldiseaseFeverUncontrolleddiabetesPregnancyHypercalcemiaRenal-compromisedpatientsHistoryoforactivePott'sdiseaseOsteomyelitisatthesurgicalsiteSepsisinoraroundthesurgicalsiteInabilitytocooperatewithand/orcomprehendpost-operativeinstructionsDEVICEINFORMATIONDBMPuttyiscomposedofDemineralizedBoneMatrixandsodiumhyaluronate.
Thedemineralizedboneallograftinthisproductispreparedfromtissueprocuredfromadeceaseddonorusingasepticsurgicaltechniques.
TheboneusedinthePuttyiscorticalbone.
ThesetissuesweretreatedwithGentamicinandwerecleanedusingethanolandwashedwithpurifiedwater.
Thebonewasdemineralizedusinghydrochloricacid.
Thedemineralizedbonewasthenlyophilizedtoacontrolledmoisturecontent.
Thedemineralizedbonewascombinedwithsterile-filteredsodiumhyaluronatepriortopackaging.
Sodiumhyaluronateisanaturallyderivedmaterialthatisbiocompatibleandbiodegradable.
ThesodiumhyaluronateismixedinaphosphatebufferedsalineandisaddedtothedemineralizedbonetoaidinmaintainingphysiologicalpHaswellastoimprovethehandlingcharacteristicsofdemineralizedbone.
Sometissuesaretreatedwithlow-dosegammaradiation.
Forthesetissuesthecontainerlabelwillstate,"TreatedwithGammaRadiation.
"SamplesfromeachdonorlotofDBMPuttyweretestedandshowednoevidenceofmicrobialgrowth,complyingwiththerequirementsofUSPSterilityTests.
THISTISSUEWASRECOVEREDFROMADECEASEDDONORFROMWHOMLEGALAUTHORIZATIONORCONSENTHASBEENOBTAINED.
THISRECOVERYWASPERFORMEDUSINGASEPTICTECHNIQUES.
PROCESSINGANDPACKAGINGWEREPERFORMEDUNDERASEPTICCONDITIONS.
TERMINALSTERILIZATIONAGENTSWERENOTUSEDINTHEPROCESS.
DBMPuttyComponentsSizeBoneParticleDiameter212–850mSodiumhyaluronatecontent(byweightinsolution)4%Bonecontent(byweight)31%INSTRUCTIONSFORUSEDBMPuttyispackagedinaglasssyringeandmustbeextrudedintoasterilebasin,notdirectlyintotheoperativesite.
THESYRINGEISNOTANAPPLICATOR.
Careshouldbetakentoapplygentle,evenforcetotheplungerwhenextrudingDBMPuttyfromthesyringe.
Extremeforceappliedtotheplungermaycausetheglasssyringetobreak.
NOTE:Thisallografthasbeenasepticallypackagedintosterilizedpackagingcomponents.
Tomakereadyforuse,openthepackageusingaseptic/steriletechniques.
1.
Peelbacklidofoutertray.
2.
Passinnertraytosterilefield.
3.
Peelbacklidofinnertray.
4.
Removesyringefrominnertray.
5.
Removeprotectivecapfromendofsyringe.
6.
ExtrudeDBMPuttyintoasterilebasin.
7.
ShapeanduseDBMPuttyaspersurgeon'spreference.
DBMPuttycanbeusedaloneormixedwithautogenousbone(1:1ratiobyvolume)orwithbonemarrowaspirate(2.
0mL/2.
8gofDBMPuttyor2.
0cc/2.
8ccofDBMPutty).
Disposeofexcessorunusedtissueandallpackagingthathasbeenincontactwiththetissueinaccordancewithrecognizedproceduresfordiscardingregulatedmedicalwastematerials.
DONORSCREENING&TESTINGPriortodonation,thedonor'smedical/socialhistoryisscreenedformedicalconditionsordiseaseprocessesthatwouldcontraindicatethedonationoftissuesinaccordancewithcurrentpoliciesandproceduresapprovedbytheMTFMedicalBoardofTrustees.
DonorbloodsamplestakenatthetimeofrecoveryweretestedbyafacilitythatisCLIAcertifiedandregisteredwiththeFDA.
Thedonorbloodsamplesweretestedfor:HepatitisBsurfaceantigenHIV-1/2antibodyHepatitisBcoreantibodySyphilisHepatitisCantibodyHBV(NAT)HIV-1(NAT)HCV(NAT)Allinfectiousdiseasetestswerenegative.
Thisallografttissuehasbeendeterminedtobesuitablefortransplantation.
Theinfectiousdiseasetestresults,consent,currentdonormedicalhistoryinterview,physicalassessment,availablerelevantmedicalrecordstoincludepreviousmedicalhistory,laboratorytestresults,autopsyandcoronerreports,ifperformed,andinformationobtainedfromanysourceorrecordswhichmaypertaintodonorsuitability,havebeenevaluatedbyanMTFphysicianandaresufficienttoindicatethatdonorsuitabilitycriteriacurrentatthetimeofprocurement,havebeenmet.
Thistissueissuitablefortransplantation.
ThedonorsuitabilitycriteriausedtoscreenthisdonorareincompliancewiththeFDAregulationspublishedin21CFRPart1271HumanCells,Tissues,andCellularandTissueBasedProducts,asapplicable.
Allproceduresfordonorscreening,serologicandmicrobiologictesting,meetorexceedcurrentstandardsestablishedbytheAmericanAssociationofTissueBanks.
VIRALCLEARANCEANDINACTIVATIONApanelofmodelpotentialhumanvirusesrepresentingvariousvirustypes,sizes,shapesandgenomeswereevaluated.
Theviralinactivationtestingdemonstratedsuitableviralinactivationpotentialoftheprocessingmethodforawidespectrumofpotentialhumanviruses.
TheDBMPuttyprocessfurtherreducestheriskofviralcontaminationbeyonddonortestingandscreeningprocedures.
PACKAGING&LABELINGDBMPuttyisasepticallypackagedinasterilizedsyringe.
ThesyringecontainingDBMPuttyisinsidetwosterilizedplastictrays,eachsealedwithfoillids.
Theoutertrayislabeledandthenputinabox.
Thisallograftmustnotbeusedunderanyofthefollowingcircumstances:Ifthecontainersealisdamagedornotintactorhasanyphysicaldamage;Ifthecontainerlabeloridentifyingbarcodeisseverelydamaged,notlegibleorismissing;orIftheexpirationdateshownonthecontainerlabelhaspassed.
STORAGEStoreDBMPuttyatambienttemperature.
Norefrigerationorfreezingisrequired.
Itistheresponsibilityofthetransplantfacilityorcliniciantomaintainthetissueintendedfortransplantationintheappropriaterecommendedstorageconditionspriortotransplant.
PATIENTRECORDTissuerecipientrecordsmustbemaintainedbytheconsigneeandtransplantfacilityforthepurposeoftracingtissuepost-transplantation.
ATissueTraceTrackingFormandpeel-offstickershavebeenincludedwitheachpackageoftissue.
Theserialnumberandthetissuedescriptionhavebeenpreprintedonthepeel-offstickers.
PleaserecordthepatientID,nameandaddressofthetransplantfacility,allografttissueinformation(usingthepeel-offstickers),andcommentsregardingtheuseofthetissueontheTissueTraceTrackingForm.
AlternativelyasystemforelectronicsubmissionmaybeusedandsenttoMTFTTC@Sceris.
com.
WithintheUnitedStates:Oncecompleted,thebottompageoftheformshouldbereturnedtoMTFusingtheself-addressedmailer.
Copiesofthisinformationshouldberetainedbythetransplantfacilityforfuturereference.
OutsideoftheUnitedStates:Oncecompleted,thebottompageoftheformshouldbereturnedtothelocalallograftrepresentativeorprovider.
Copiesofthisinformationshouldberetainedbythehospitalforfuturereference.
Reference:CurrentMTFpoliciesandproceduresareincompliancewithcurrentFDA,AATBandotherregulatoryrequirements.
DefinitionsofLabelSymbolsSeeIFUDoNotReuseProcessedby:125MayStreetEdison,NJ08837USAWithintheUnitedStates:1.
800.
433.
6576OutsidetheUnitedStates:+1.
732.
661.
0202Allrecovery,processinganddistributioncostswerepaidforbyMTF,anon-profitorganization.
CAUTION:Federal(US)lawrestrictsthisdevicetosale,distributionandusebyorontheorderofaphysicianordentist.
Pleasenote:Humantissuefortransplantationshallnotbeoffered,distributedordispensedforVeterinaryUse.
MTFtissueformsandproductsareprotectedbyoneormoreissuedorlicensedUnitedStatespatents.
AlistofpatentsonavailabletissuesandrelatedtechnologiesmaybefoundontheMTFwebsitewww.
mtf.
org.
MTFMusculoskeletalTransplantFoundationandTissueTraceareregisteredtrademarksoftheMusculoskeletalTransplantFoundation,Edison,NJUSA.
2016MusculoskeletalTransplantFoundation.
CTO:100024
AsepticallyProcessed.
PassesUSPSterilityTests.
DBMPuttyIsNotTerminallySterilized.
DESCRIPTIONDBMPuttyisprocessedhumanbonethathasbeendemineralizedandcombinedwithsodiumhyaluronate,whichisanaturallyderivedmaterialnotofanimaloriginthatisbothbiocompatibleandbiodegradable.
Thecombinationofdemineralizedboneandsodiumhyaluronateresultsinaputty-likeortexturedconsistencyforeaseandflexibilityofuseduringsurgicalapplication.
OSTEOINDUCTIVEPOTENTIALDBMPuttyisosteoconductive,andhasbeenshowntohaveosteoinductivepotentialinanathymicmousemodel.
EverylotoffinalDBMPuttyproductistestedinanathymicmousemodelorinanalkalinephosphataseassay,whichhasbeenshowntohaveapositivecorrelationwiththeathymicmousemodel,toensuretheosteoinductivepotentialofthefinalproduct.
Standardtestingperformedinanathymicmouseoralkalinephosphataseassaymustprovepositiveforlotrelease.
Itisunknownhowtheosteoinductivepotential,measuredintheathymicmousemodelorthealkalinephosphataseassay,willcorrelatewithclinicalperformanceinhumansubjects.
INDICATIONSFORUSEDBMPuttyisintendedfortheaugmentationofdeficientmaxillaryandmandibularalveolarridgesandthetreatmentoforal/maxillofacialanddentalintraosseousdefectsincluding:-Ridgeaugmentation-Fillingofcysticdefect-Fillingofextractionsites-Fillingoflesionsofperiodontalorigin-Craniofacialaugmentation-FillingofdefectsofEndodonticorigin-Mandibularreconstruction-Repairoftraumaticdefectsofthealveolarridge,excludingmaxillaryandmandibularfracture-Fillingresectiondefectsinbenignbonetumors,benigncystsorotherosseousdefectsinthealveolarridgewallDBMPuttycanbeusedwithautograftorbonemarrowaspirate.
DBMPuttyisforsinglepatientuseonly.
CONTRAINDICATIONSDBMPuttyisNOTintendedtoprovidestructuralsupportoftheboneduringthehealingprocess.
DBMPuttyisalsocontraindicatedforincompletemaxillofacialskullgrowth.
ADVERSEEFFECTSPossibleadverseeffectsofusingDBMPuttyinclude,butarenotlimitedto:PotentiallossofcontourofmaxillofacialskullInfectionofsofttissueand/orbone(osteomyelitis)FeverDeformityoftheboneatthesiteIncompleteboneingrowth,delayedunionornon-unionHypercalcemiaortransienthypercalcemiaFractureofthenewlyformedboneDiseasetransmissionandundesirableimmuneresponseWithintheUnitedStates:AdverseoutcomesattributabletothetissuemustbepromptlyreportedtoMTF.
OutsideoftheUnitedStates:Adverseoutcomesattributabletothetissuemustbepromptlyreportedtoyourlocalrepresentative.
CAUTIONSDonotsterilize.
Donotfreeze.
DBMPuttymayextrudeintofacialsofttissue.
TraceamountsofGentamicinmaybepresent.
Tissueisexposedtoprocessingsolutionsthatmaycontaindetergentsandalcohol.
Traceamountsofprocessingsolutionmayremain.
Cautionshouldbeexercisedifthepatientisallergictoanyofthesesubstances.
NOTE:No-lactamantibioticsareusedduringtheprocessingoftissueinDBMPuttyproducts.
ExtensivemedicalscreeningprocedureshavebeenusedintheselectionofalltissuedonorsforMTF(pleaseseeDonorScreeningandTesting).
TransmissionofinfectiousdiseasessuchasHIVorhepatitis,aswellasatheoreticalriskoftheCreutzfeldt-Jakob(CJD)agent,mayoccurinspiteofcarefuldonorselectionandserologicaltesting.
Closedsuctionordrainageisrecommendedtopreventfluidaccumulationinthewound.
Cautionshouldbetakenforthefollowingcircumstances:SeverevascularorneurologicaldiseaseFeverUncontrolleddiabetesPregnancyHypercalcemiaRenal-compromisedpatientsHistoryoforactivePott'sdiseaseOsteomyelitisatthesurgicalsiteSepsisinoraroundthesurgicalsiteInabilitytocooperatewithand/orcomprehendpost-operativeinstructionsDEVICEINFORMATIONDBMPuttyiscomposedofDemineralizedBoneMatrixandsodiumhyaluronate.
Thedemineralizedboneallograftinthisproductispreparedfromtissueprocuredfromadeceaseddonorusingasepticsurgicaltechniques.
TheboneusedinthePuttyiscorticalbone.
ThesetissuesweretreatedwithGentamicinandwerecleanedusingethanolandwashedwithpurifiedwater.
Thebonewasdemineralizedusinghydrochloricacid.
Thedemineralizedbonewasthenlyophilizedtoacontrolledmoisturecontent.
Thedemineralizedbonewascombinedwithsterile-filteredsodiumhyaluronatepriortopackaging.
Sodiumhyaluronateisanaturallyderivedmaterialthatisbiocompatibleandbiodegradable.
ThesodiumhyaluronateismixedinaphosphatebufferedsalineandisaddedtothedemineralizedbonetoaidinmaintainingphysiologicalpHaswellastoimprovethehandlingcharacteristicsofdemineralizedbone.
Sometissuesaretreatedwithlow-dosegammaradiation.
Forthesetissuesthecontainerlabelwillstate,"TreatedwithGammaRadiation.
"SamplesfromeachdonorlotofDBMPuttyweretestedandshowednoevidenceofmicrobialgrowth,complyingwiththerequirementsofUSPSterilityTests.
THISTISSUEWASRECOVEREDFROMADECEASEDDONORFROMWHOMLEGALAUTHORIZATIONORCONSENTHASBEENOBTAINED.
THISRECOVERYWASPERFORMEDUSINGASEPTICTECHNIQUES.
PROCESSINGANDPACKAGINGWEREPERFORMEDUNDERASEPTICCONDITIONS.
TERMINALSTERILIZATIONAGENTSWERENOTUSEDINTHEPROCESS.
DBMPuttyComponentsSizeBoneParticleDiameter212–850mSodiumhyaluronatecontent(byweightinsolution)4%Bonecontent(byweight)31%INSTRUCTIONSFORUSEDBMPuttyispackagedinaglasssyringeandmustbeextrudedintoasterilebasin,notdirectlyintotheoperativesite.
THESYRINGEISNOTANAPPLICATOR.
Careshouldbetakentoapplygentle,evenforcetotheplungerwhenextrudingDBMPuttyfromthesyringe.
Extremeforceappliedtotheplungermaycausetheglasssyringetobreak.
NOTE:Thisallografthasbeenasepticallypackagedintosterilizedpackagingcomponents.
Tomakereadyforuse,openthepackageusingaseptic/steriletechniques.
1.
Peelbacklidofoutertray.
2.
Passinnertraytosterilefield.
3.
Peelbacklidofinnertray.
4.
Removesyringefrominnertray.
5.
Removeprotectivecapfromendofsyringe.
6.
ExtrudeDBMPuttyintoasterilebasin.
7.
ShapeanduseDBMPuttyaspersurgeon'spreference.
DBMPuttycanbeusedaloneormixedwithautogenousbone(1:1ratiobyvolume)orwithbonemarrowaspirate(2.
0mL/2.
8gofDBMPuttyor2.
0cc/2.
8ccofDBMPutty).
Disposeofexcessorunusedtissueandallpackagingthathasbeenincontactwiththetissueinaccordancewithrecognizedproceduresfordiscardingregulatedmedicalwastematerials.
DONORSCREENING&TESTINGPriortodonation,thedonor'smedical/socialhistoryisscreenedformedicalconditionsordiseaseprocessesthatwouldcontraindicatethedonationoftissuesinaccordancewithcurrentpoliciesandproceduresapprovedbytheMTFMedicalBoardofTrustees.
DonorbloodsamplestakenatthetimeofrecoveryweretestedbyafacilitythatisCLIAcertifiedandregisteredwiththeFDA.
Thedonorbloodsamplesweretestedfor:HepatitisBsurfaceantigenHIV-1/2antibodyHepatitisBcoreantibodySyphilisHepatitisCantibodyHBV(NAT)HIV-1(NAT)HCV(NAT)Allinfectiousdiseasetestswerenegative.
Thisallografttissuehasbeendeterminedtobesuitablefortransplantation.
Theinfectiousdiseasetestresults,consent,currentdonormedicalhistoryinterview,physicalassessment,availablerelevantmedicalrecordstoincludepreviousmedicalhistory,laboratorytestresults,autopsyandcoronerreports,ifperformed,andinformationobtainedfromanysourceorrecordswhichmaypertaintodonorsuitability,havebeenevaluatedbyanMTFphysicianandaresufficienttoindicatethatdonorsuitabilitycriteriacurrentatthetimeofprocurement,havebeenmet.
Thistissueissuitablefortransplantation.
ThedonorsuitabilitycriteriausedtoscreenthisdonorareincompliancewiththeFDAregulationspublishedin21CFRPart1271HumanCells,Tissues,andCellularandTissueBasedProducts,asapplicable.
Allproceduresfordonorscreening,serologicandmicrobiologictesting,meetorexceedcurrentstandardsestablishedbytheAmericanAssociationofTissueBanks.
VIRALCLEARANCEANDINACTIVATIONApanelofmodelpotentialhumanvirusesrepresentingvariousvirustypes,sizes,shapesandgenomeswereevaluated.
Theviralinactivationtestingdemonstratedsuitableviralinactivationpotentialoftheprocessingmethodforawidespectrumofpotentialhumanviruses.
TheDBMPuttyprocessfurtherreducestheriskofviralcontaminationbeyonddonortestingandscreeningprocedures.
PACKAGING&LABELINGDBMPuttyisasepticallypackagedinasterilizedsyringe.
ThesyringecontainingDBMPuttyisinsidetwosterilizedplastictrays,eachsealedwithfoillids.
Theoutertrayislabeledandthenputinabox.
Thisallograftmustnotbeusedunderanyofthefollowingcircumstances:Ifthecontainersealisdamagedornotintactorhasanyphysicaldamage;Ifthecontainerlabeloridentifyingbarcodeisseverelydamaged,notlegibleorismissing;orIftheexpirationdateshownonthecontainerlabelhaspassed.
STORAGEStoreDBMPuttyatambienttemperature.
Norefrigerationorfreezingisrequired.
Itistheresponsibilityofthetransplantfacilityorcliniciantomaintainthetissueintendedfortransplantationintheappropriaterecommendedstorageconditionspriortotransplant.
PATIENTRECORDTissuerecipientrecordsmustbemaintainedbytheconsigneeandtransplantfacilityforthepurposeoftracingtissuepost-transplantation.
ATissueTraceTrackingFormandpeel-offstickershavebeenincludedwitheachpackageoftissue.
Theserialnumberandthetissuedescriptionhavebeenpreprintedonthepeel-offstickers.
PleaserecordthepatientID,nameandaddressofthetransplantfacility,allografttissueinformation(usingthepeel-offstickers),andcommentsregardingtheuseofthetissueontheTissueTraceTrackingForm.
AlternativelyasystemforelectronicsubmissionmaybeusedandsenttoMTFTTC@Sceris.
com.
WithintheUnitedStates:Oncecompleted,thebottompageoftheformshouldbereturnedtoMTFusingtheself-addressedmailer.
Copiesofthisinformationshouldberetainedbythetransplantfacilityforfuturereference.
OutsideoftheUnitedStates:Oncecompleted,thebottompageoftheformshouldbereturnedtothelocalallograftrepresentativeorprovider.
Copiesofthisinformationshouldberetainedbythehospitalforfuturereference.
Reference:CurrentMTFpoliciesandproceduresareincompliancewithcurrentFDA,AATBandotherregulatoryrequirements.
DefinitionsofLabelSymbolsSeeIFUDoNotReuseProcessedby:125MayStreetEdison,NJ08837USAWithintheUnitedStates:1.
800.
433.
6576OutsidetheUnitedStates:+1.
732.
661.
0202Allrecovery,processinganddistributioncostswerepaidforbyMTF,anon-profitorganization.
CAUTION:Federal(US)lawrestrictsthisdevicetosale,distributionandusebyorontheorderofaphysicianordentist.
Pleasenote:Humantissuefortransplantationshallnotbeoffered,distributedordispensedforVeterinaryUse.
MTFtissueformsandproductsareprotectedbyoneormoreissuedorlicensedUnitedStatespatents.
AlistofpatentsonavailabletissuesandrelatedtechnologiesmaybefoundontheMTFwebsitewww.
mtf.
org.
MTFMusculoskeletalTransplantFoundationandTissueTraceareregisteredtrademarksoftheMusculoskeletalTransplantFoundation,Edison,NJUSA.
2016MusculoskeletalTransplantFoundation.
CTO:100024
- Transmissionputty相关文档
- REPLputty
- 皮肤putty
- utilityputty
- jeffputty
- 思科putty
- dependingputty
麻花云:3折优惠,香港CN2安徽麻花云香港安徽移动BGP云服务器(大带宽)
麻花云在7月特意为主机测评用户群定制了促销活动:香港宽频CN2云服务器、安徽移动云服务器(BGP网络,非单线,效果更好)、安徽移动独立服务器、安徽电信独立服务器,全部不限制流量,自带一个IPv4,默认5Gbps的DDoS防御。活动链接:https://www.mhyun.net/act/zjcp特价云服务器不限流量,自带一个IPv4,5Gbps防御香港宽频CN2全固态Ⅲ型 4核4G【KVM】内存:...

易探云330元/年,成都4核8G/200G硬盘/15M带宽,仅1888元/3年起
易探云服务器怎么样?易探云是国内一家云计算服务商家,致力香港云服务器、美国云服务器、国内外服务器租用及托管等互联网业务,目前主要地区为运作香港BGP、香港CN2、广东、北京、深圳等地区。目前,易探云推出的国内云服务器优惠活动,国内云服务器2核2G5M云服务器低至330元/年起;成都4核8G/200G硬盘/15M带宽,仅1888元/3年起!易探云便宜vps服务器配置推荐:易探云vps云主机,入门型云...
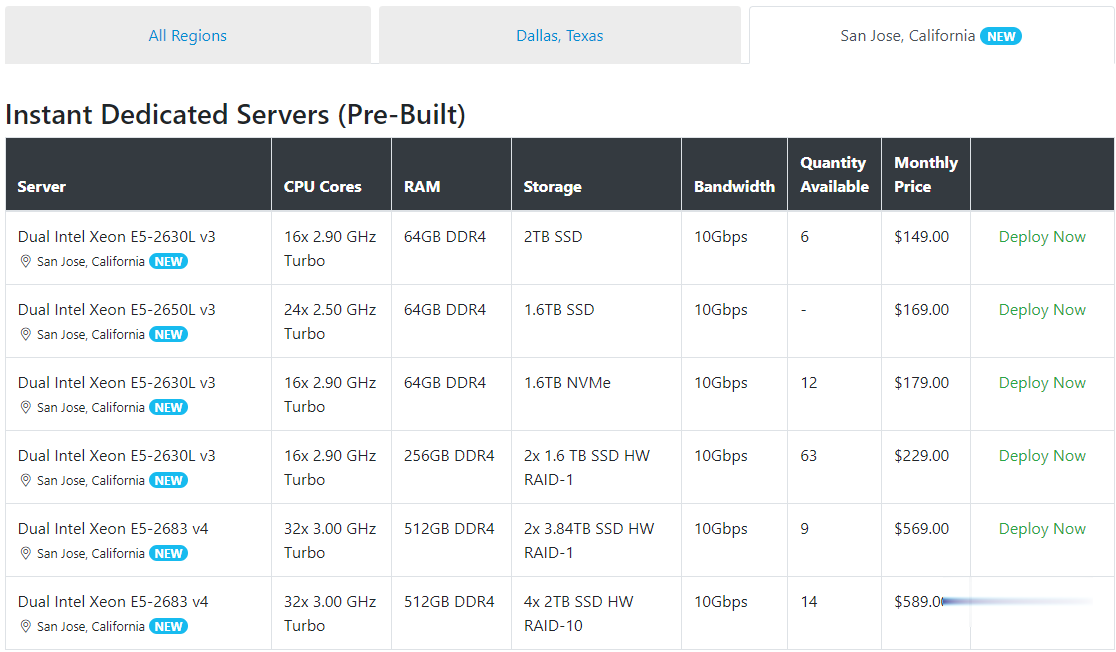
Spinservers美国圣何塞服务器$111/月流量10TB
Spinservers是Majestic Hosting Solutions,LLC旗下站点,主营美国独立服务器租用和Hybrid Dedicated等,数据中心位于美国德克萨斯州达拉斯和加利福尼亚圣何塞机房。TheServerStore.com,自 1994 年以来,它是一家成熟的企业 IT 设备供应商,专门从事二手服务器和工作站业务,在德克萨斯州拥有 40,000 平方英尺的仓库,库存中始终有...
putty为你推荐
-
iphone6s内存多大iPhone6S内存多大?16G够用吗名侦探柯南644名侦探柯南中有铃木园子出现的是哪几集?法兰绒和珊瑚绒哪个好请问天鹅绒法兰绒和珊瑚绒哪个好啊 想买套床上用品闪迪和金士顿哪个好tf卡闪迪和金士顿哪个更好音乐播放器哪个好最好的音乐播放器下载等额本息等额本金哪个好等额本金和等额本息哪个划算?如果想在5-10年内还清贷款哪类更划算一些?红茶和绿茶哪个好红茶和绿茶 那个更好视频软件哪个好手机看视频用什么软件好空间登录页面怎样用网页登录到自己的QQ空间?辽宁联通网上营业厅网中国联通的初始服务密码一般是多少