COPD103838.com
103838.com 时间:2021-03-19 阅读:()
JNeurol(2017)264:2457–2463DOI10.
1007/s00415-017-8647-0ORIGINALCOMMUNICATIONAnopenlabelstudytoassessthefeasibilityandtolerabilityofrilmenidineforthetreatmentofHuntington'sdiseaseBenjaminR.
Underwood1·ZeynW.
GreenThompson2·PeterJ.
Pugh2·StanleyE.
Lazic3·SarahL.
Mason7·JulesGriffin4·P.
SimonJones5·JamesB.
Rowe5·DavidC.
Rubinsztein6,7·RogerA.
Barker2,5,8Received:11September2017/Revised:9October2017/Accepted:11October2017/Publishedonline:26October2017TheAuthor(s)2017.
Thisarticleisanopenaccesspublicationindisease-specificvariables,suchasmotor,cognitiveandfunctionalperformance,structuralMRIandserummetabo-lomicanalysis.
12patientscompletedthestudy;reasonsforwithdrawalincludedproblemstoleratingstudyprocedures(MRI,andvenepuncture),depressionrequiringhospitaladmissionandlogisticalreasons.
Threeseriousadverseeventswererecorded,includinghospitalisationfordepres-sion,butnonewerethoughttobedrug-related.
Changesinsecondaryoutcomeswereanalysedastheannualrateofchangeinthestudygroup.
Theoverallchangewascom-parabletochangesseeninrecentlargeobservationalstud-iesinHDpatients,thoughdirectstatisticalcomparisonstothesestudieswerenotmade.
Chronicoraladministrationofrilmenidineisfeasibleandwell-toleratedandfuture,larger,placebo-controlled,studiesinHDarewarranted.
Trialregistration:EudraCTnumber2009-018119-14.
KeywordsAutophagy·Neurodegenerativedisease·Interventionaltrial·MRI·CognitionIntroductionHuntington'sdisease(HD)isaneurodegenerativedisorderwhichpresentswithacombinationofmovement,psychi-atricandcognitivedeficits(reviewedinBatesetal.
[1]).
Ittypicallyprogresseswithincreasingdisabilitytodeathoverthecourseof15–20years.
Therearecurrentlynodisease-modifyingtreatmentsavailablewhichcanaltertheprogres-sionofHD[2].
Fromtheveryfirstdescription,itwasrecognisedthattheconditionisfamilial,andtheunderlyinggeneticmutation,anexpandedtrinucleotiderepeatintheHuntingtingeneontheshortarmofchromosomefour,wasidentifiedin1993[3].
ThishasledtoabetterunderstandingofthemolecularAbstractPreclinicaldatahaveshownthatrilmenidinecanregulateautophagyinmodelsofHuntington'sdisease(HD),providingapotentialroutetoalterthediseasecourseinpatients.
Consequently,a2-yearopen-labelstudyexam-iningthetolerabilityandfeasibilityofrilmenidineinmild-moderateHDwasundertaken.
18non-dementedpatientswithmildtomoderateHDtookdailydosesof1mgRilme-nidinefor6monthsand2mgforafurther18monthsfol-lowedbya3-monthwashoutperiod.
Theprimaryoutcomewasthenumberofwithdrawalsandseriousadverseevents.
SecondaryoutcomesincludedsafetyparametersandchangesElectronicsupplementarymaterialTheonlineversionofthisarticle(doi:10.
1007/s00415-017-8647-0)containssupplementarymaterial,whichisavailabletoauthorizedusers.
*RogerA.
Barkerrab46@cam.
ac.
uk1FulbournHospital,Fulbourn,CambridgeCB215EF,UK2Addenbrooke'sHospital,HillsRoad,CambridgeCB212QQ,UK3QuantitativeBiology,DiscoverySciences,IMEDBiotechUnit,AstraZeneca,CambridgeCB40WG,UK4DepartmentofBiochemistry,UniversityofCambridge,CambridgeCB21GA,UK5DepartmentofClinicalNeurosciences,andMRCCognitionandBrainSciencesUnit,UniversityofCambridge,CambridgeBiomedicalCampus,CambridgeCB20SZ,UK6CambridgeInstituteforMedicalResearch,UniversityofCambridge,CambridgeBiomedicalCampus,HillsRoad,CambridgeCB20XY,UK7UKDementiaResearchInstitute,UniversityofCambridge,HillsRoad,CambridgeCB20XY,UK8JohnVanGeestCentreforBrainRepair,ForvieSite,RobinsonWay,CambridgeCB20PY,UK2458JNeurol(2017)264:2457–2463pathophysiologyofthecondition,andconsequentlytorationaltreatmentapproaches.
Oneexampleofthishasbeentherecog-nitionthatthemutantproteinproducedbythegeneticmutationisasubstratefordegradationbyautophagy[4].
Autophagydescribesaprocesswhereintracytoplasmicves-icles,calledautophagosomes,delivercytoplasmiccontentstolysosomesfordegradation.
Itisamenabletoup-regulationbydrugsinavarietyofcellular,fly,zebrafishandmousemodelsofHD,andthisstrategyamelioratessignsofthediseaseinthesemodels.
Theoriginaldrugthatwasshowntodothiswasrapamycin,aninhibitorofthemammaliantargetofrapamycinormTOR[5].
However,whilethisdrugisapprovedforhumanuse,ithasasignificantsideeffectprofileincludingimmu-nosuppressionmakingitanunattractiveoptionfortriallinginpatientswithchronicneurodegenerativedisordersoftheCNS.
Subsequentdrugscreenshaveidentifiedlesstoxicup-regulatorsofautophagywhichstillretaintheabilitytorescuecellsandbehaviourinmammalianandothermodelsofHD[6].
Onesuchdrugisrilmenidine(N-(dicyclopropylmethyl)-4,5-dihydro-2-oxazolamine),anα2receptorantagonistandanimidazolineI1receptoragonist[7].
Ithasbeenextensivelyusedintheclinicasacentrallyactinganti-hypertensiveagentwithnosignificantsideeffectscomparedtoplaceboatdosesof1mgperdayandwithanextensivesafetyrecordinhumanuseincludingelderlysubjects.
Thus,thisagentisanidealcandidatetoassessforthefeasibilityandtolerabilityofthisapproachinpatientswithHD.
Investigatingwhethersuchtreatmentscanreallyslowdiseaseprogressioninchronicneurodegenerativedisorders,suchasHD,willrequirelargeexpensivestudies.
Indeed,longitudinalobservationalstudiesofpatientswithHunting-ton'sdiseaseover2–3years(forexample,COHORT[8]andTRACK-HD[9–12])havehighlightedthatdefinitivelystudyingtheeffectsofdisease-modifyingagentsonHDwillrequirelargenumbersofpatientsfollowedforseveralyears.
However,beforeembarkingonstudiesofthistype,itisimportanttoascertainthatthedrugtobetriallediswelltoleratedandthestudyfeasible.
WethereforeundertookafirstinHDsmallopen-labelstudyovera2-yearperiodlookingatthetolerabilityandfeasibilityofrilmenidineasapossibledisease-modifyingagent.
Whilenoaccountofitseffectivenesscanbecon-cludedfromsuchastudy,ithasprovidedencouragingdatawhichcannowbeusedtoplanmoredefinitivelarge-scale,multi-centre,double-blind,placebocontroltrials.
MethodsTrialdesignandparticipantsThiswasasinglecentreopen-labelstudycarriedoutwithpatientsrecruitedfromtheregionalHDclinicattheCambridgeUniversityHospitalsNHSTrust(Addenbrooke'sHospital),UK.
Writteninformedconsentwasobtainedfromallparticipants.
Inclusioncriteria1.
AconfirmeddiagnosisofHuntington'sdiseaseonthebasisofqualifyingclinicalsignsandsymptoms,spe-cificallyaUnifiedHuntington'sDiseaseRatingScale1999(UHDRS)totalmotorscoreofatleast5[9],andconfirmationofaCAGrepeatof>36inexon1ofthehttgene.
2.
HDstage2–3asdefinedusingaUHDRStotalfunc-tionalcapacity(TFC)scoreofgreaterthan4[13].
3.
Ambulantandabletoself-careindependently.
4.
Agedbetween18and70.
5.
Englishspeakingandabletogivewritten,informedcon-sent.
Exclusioncriteria1.
Anongoingclinicallysignificantandunstablegen-eralmedicalcondition[includingbutnotlimitedto;asthma,chronicobstructivepulmonarydisease(COPD),ongoingischaemicheartdiseaseproblems(IHD),congestivecardiacfailure(CCF),leftbundlebranchblock(LBBB)oracerebrovascularaccident(CVA)]confirmedviapastmedicalhistoryorbaselinemedicalorphysicalexaminationandinvestigations.
2.
Prescribedanti-hypertensivemedicationoranydrugknowntobecontraindicatedortohaveanadverseinteractionwithrilmenidine(viz.
amonoamineoxi-daseinhibitor).
3.
Knownhypersensitivitytorilmenidine.
4.
Ongoingsignificantmentalillnessdeterminedbyevi-dence,orahistory,ofapsychoticoraffective(depres-sionormania)episodeinthe6monthspriortobase-lineInterviewasassessedusingtheDiagnosticandStatisticalManualofMentalDisorderscriteria(FourthEditionwithTextRevision;AmericanPsychiatricAssociation).
5.
Prescribedtypicaloratypicalanti-psychoticmedica-tionwhenbeingexplicitlyusedtotreatapsychoticillness(asopposedtothemovementdisorderofHD).
6.
Pregnantorbreastfeedingfemalepatients,includingthoseplanningtoconceiveduringtheperiodofthetrial.
Womenofchildbearingagewhowereneitherpregnantnorplanningtoconceiveduringtheperiodofthestudyweredeemedeligibleprovidedtheyusedtwoformsofcontraception,atleastoneofwhichhadtobeabarriermethod.
2459JNeurol(2017)264:2457–24637.
Substance(alcoholorillegal/prescriptiondrug)misuseinthe6monthspriortothebaselineassessment.
8.
Knownco-morbidmajorneurologicaldisorder(includ-ingParkinson'sdiseaseoranestablisheddementia),HIV/AIDSorhepatitis(HBVorHCV).
9.
Previousneurosurgerytothebrain.
10.
Markedclinicallyadverseabnormalitiesonlaboratoryinvestigationsincludingcreatinineclearance177Umol/l.
InterventionsandoutcomesAllpatientsweregiven1mgrilmenidinedailyfor6monthsandthen2mgdailyforthenext18monthsofthestudy.
Followingscreening,patientswereseenforabaselineassessmentandthenreviewedatmonths1,3,6,9,12,18,24and27(i.
e.
3monthsafterstop-pingtherilmenidine).
Seriousadverseevents(SAEs)andwithdrawals(allprimaryendpoints)wererecordedalongwithnon-seriousadverseevents,height,weight,vitalsigns,routinehaematologyandbiochemistrybloodmeasuresandECGoccurredasperthescheduleofevents(Table1).
SpecificmeasuresoftheprogressionoftheirHDincludedUHDRStotalmotorscore,functionalandindependencescales[13]aswellastheirperformanceonthetrailmak-ingtest,MiniMentalStateExamination(MMSE)[14]andverbalfluencytasks(asassessedbythecontrolledoralwordassociationtest,COWAT).
OthercognitivetestsfromtheCambridgeAutomatedNeuropsychologicalTestAutomatedBattery(CANTAB)[15]werealsousedandincludedthemeantimetaken(latency)andtotalnumberofcorrectlysolvedproblemsontheOneTouchStockingsofCambridge,andthetotalnum-beroferrorsmadepriortoandattheextra-dimensionalshiftstageoftheCANTABintra-dimensional/extra-dimensionalset-shiftingtask(ID/ED)[15]).
ThescheduleofeventsisshowninTable1.
SamplesizeandstatisticalmethodsTheestimateoftherequiredsamplesize(n=16)wasdesignedtodetectSAEs/AEsratherthanasignificantchangeinsecondaryendpointsandwasbasedondatagatheredfromourpreviousstudiesusingmetabolomicbiomarkersinHDpatients[16].
Thenumberofseriousadverseeventsanddropoutswasanalysedwithanexactbinomialtest,testingwhethertheratesweregreaterthananacceptablesafetylevelof5%peryear,or10%overthecourseofthestudy.
One-sidedpvaluesand95%CIarereported.
Forsecondaryoutcomes,themeanrateofprogression(slope)wasthemainparameterofinterest.
Theseoutcomeswereanalysedwithmultilevelmodelsallowingforpatient-specificinterceptsandslopes.
Thebasicmodelforthesec-ondaryanalyseswaswhereyijistheoutcomeforpatientiattimej,andαpatientandβpatientarethepatient-specificinterceptsandslopes,respec-tively.
Priorsformodelparameterswerenon-informative(oryijNormal(patient[i]+patient[i]timeij,),Table1AssessmentscheduleforparticipantsinthetrialBaselineOnemonthreviewMonths3,6,9,12,18and2427monthsMedicalhistory,familyhistoryandphysicalexaminationXXxXWeightXXxXVitalsigns(bloodpressureandpulse/heartrate)XXxXHaematologyXXxXBiochemistryXXxX12-LeadECGXXxXReviewofadverseeventsXXxXBloodsampleformetabolomicsXxXReviewofconcomitantmedicationsXxXMagneticresonanceimaging(brain)XXUnifiedHuntington'sDiseaseRatingScaleXxXMinimentalStateExaminationXxXNeuropsychiatricInventoryXxxTrailmakingtestXxXVerbalfluencyXxXTowerofLondon(OneTouchStockingsofCambridge)XxXExtra-dimensional/intra-dimensionalshiftsXxX2460JNeurol(2017)264:2457–2463verymildlyinformativetoaidconvergence).
Forpositivelyskewedoutcomes,thedatawerelog-transformedandsomeoutcomesusedabinomialornegativebinomiallikelihoodifthedatawasbettermodelledasproportionsorcounts.
Theresultsarepresentedastheannualrateofchangeandtheconfidenceintervalsarethe95%highestposteriordensityintervals.
pvaluesrepresenttheproportionoftheposte-riordensityontheoppositesideofzerofromtheestimatedmean.
Forexample,theUHDRSmotorscoreincreasedby3.
5unitsannually,andtheassociatedpvalueof0.
0063tellsusthat0.
63%oftheposteriordistributionhadnegativevalues.
ThedatawereanalysedwiththeBayesiansoftware"Stan"andtherstanandrethinkingRpackages[17,18].
Giventhelargenumberofsecondaryvariablesandthelackofdirecthypothesistesting,nostatisticaladjustmenthasbeenmadeformultipletesting.
Imaging16patientsunderwentstructuralMRimagingattheCambridgeBiomedicalCampususinga1.
5TGEMedi-calSystemsDiscoveryMR450scanner.
StructuralMRIscanswerecollectedpriortocommencementoftreatmentandat27months(i.
e.
3monthsaftertreatmentceased)toavoidanyeffectsofthemedicationonbrainvolumesandfluidshifts,givenitsknownanti-hypertensiveactions.
AT1-weighted3DBravofastspoiledgradientecho(SPGR)imagewasacquiredwithrepetitiontime(TR)=8156ms,echotime(TE)=3.
18ms,matrix=256*256,in-planeresolutionof1*1mm,252slicesof1mmthickness,inver-siontime=900msandflipangle=9°.
AnalysisoftheMRIdatawasperformedusingFreesurferv.
6(stable-v6-beta-20151015).
Theregionofinterest(ROI)valueswasimportedintoSPSS.
Apairedttestwasusedtoestimatethechangesinvolumebetweenthetwotime-points.
BloodsamplingandmetabolomicanalysisBloodsamplingformetabolomicanalysisbygaschromatog-raphy–massspectrometrywasalsoundertakenusingtech-niquespreviouslydescribed[16].
Metabolomicanalysisisatechniquewhichprovidesmultivariatequantitativeanalysisofmolecularfragments.
Measuringthepresenceofalargenumberofthesefragmentsandtheirrelativeabundanceleadstoamolecularprofilewhichcanhelpdifferentiatebetweendiseasestatesandmayalsobeusefulasapotentialbiomarkerofprogression.
ThereisreasontobelievethatmetabolismisalteredinHDandanumberofpapershavenowdescribedmetabolomicchangesinHD,includingfromourowngroup[16].
Inthisstudy,weperformedsequen-tiallongitudinalmetabolomicanalysisineachpatientasanexploratorypotentialbiomarkerofdiseaseprogression.
ResultsEighteenpatientswererecruitedtothestudy.
OnedroppedoutbeforethetreatmentwasstartedasthepatientwasunabletotolerateMRIscanning.
Afurtherindividualdroppedoutatthe1-monthvisitasthepatientwasunabletotoleratevenepuncture.
Thisleft16patientsforwhomthereweremorethanoneobservationandwhoformedthetrialpopulation.
Onepatientwithdrewfromthestudyafter6monthswhentheybecamedepressedandrequiredpsychiatricadmission.
Onefurtherpatientdroppedoutat21monthsasthepatientwasnolongerabletotraveltoclinicandtwoafterthe24-monthvisit,onewhonolongerwishedtocontinueinthetrialandtheotherwaslosttofol-low-up.
Thus,12patientscompletedthe27-monthfollow-upvisit.
ExcessivemovementartefactsintheT1imageofthefollow-upvisitofoneparticipantmeantthat11patientswereincludedinthelongitudinalimagingstudy.
ThisissummarisedinFig.
1.
Screenedn=18Excluded-unabletotolerateMRIn=1Receivedintervenonn=17Withdrewn=1Completed6monthfollow-upn=16Withdrewn=2Completed24monthfollow-upn=14Fig.
1Patientflowthroughthestudy2461JNeurol(2017)264:2457–2463RecruitmentRecruitmentoccurredbetweenOctober2012andJuly2013.
Thefinalpatientfinalvisit(FPFV)wascompletedinJune2015.
BaselinedataBaselinedemographicandclinicalcharacteristicsareshowninTable2.
PrimaryoutcomemeasuresOfthe18patientswhowererecruitedtothestudy,6with-drewforthereasonsgivenabove.
Thenumberofwith-drawalswasgreaterthanthepre-specifiedtargetrateof0.
1(p=0.
0002,6/18patients=0.
33dropoutrate;95%CI0.
16–1.
0).
However,noneofthewithdrawalswereconsid-eredtobedrug-related.
Threeseriousadverseevents(SAEs)werereported:onebrokenwristfromaphysicalfall,oneadmissiontohospitalformigraineaftertheindividualwithdrewtheirmigraineprophylaxistreatment,andoneadmissionfordepressionfol-lowingcessationofantidepressants.
Nonewerethoughttobedrug-related.
SecondaryoutcomemeasuresThesecondaryoutcomemeasuresaresummarisedinTable3(secondaryoutcomemeasures)andTable4(changesinregionalbrainvolumeonMRI).
Ofthesec-ondaryendpoints,theUHDRStotalmotorscore(+3.
5,95%CI0.
8–6.
3,p=0.
009),wholebrainvolume(1.
3%,p=0.
001)andUHDRSfunctionalcapacity(0.
4,95%CI0.
8–0.
02,p=0.
020)showedadeclineoverthestudyperiodasexpected.
Intermsofcognitivetests,latencytocorrecttimeontheCANTABOneTouchStockingsofCambridgeimproved(0.
12,95%CI0.
23to0.
01,p=0.
014),withnosig-nificantchangesinanyoftheothercognitivetests.
TheMRIdatarevealedadeclineintotalbrainvolume(excludingventricles)of1.
8%peryear(p=0.
001),althoughwithrespecttothebasalganglia,onlytheleftputa-menshowedasignificantdecreaseinsizeoverthecourseofthestudy.
Othersecondaryendpointsdidnotsignificantlychangeoverthestudy.
AllotherendpointsremainedunchangedincludingthemetabolomicanalysiswherenosignificantdifferenceswerenotedbetweenbaselineandtheendofstudyincludinginTable2BaselinedemographicandclinicalcharacteristicsBMIbodymassindex,MMSEMiniMentalStateExam,UHDRSUni-fiedHuntington'sDiseaseRatingScaleSex11male5femaleAgeMean53(range37–69)Weight80.
2kg(62.
5–100)BMI27.
5(21.
4–33.
4)MMSE27.
4(24–30)Verbalfluency23.
2(6–53)UHDRSmotor24.
5(7–46)UHDRSfunctionalcapacity9.
62(5–13)UHDRSfunctionalassessment28.
2(25–35)UHDRSindependence80(70–100)Table3Secondaryoutcomemeasures(orderedbysignificanceofthepvalue)CANTABOneTouchStockingsofCambridgeEDextra-dimensionalshiftontheCANTABintra-dimensional/extra-dimensionalset-shiftingtask,MMSEMiniMentalStateExamination,UHDRSUnifiedHuntington'sDiseaseRatingScaleVariableUnitsChangeperannumLowerCIUpperCIpvalueUHDRS(motor)Score3.
50.
86.
30.
009OTS-latencytocorrectLogs0.
120.
230.
010.
014UHDRS(totalfunctionalcapacity)Score0.
40.
80.
020.
020EDSerrorsNumber0.
20.
50.
060.
050UHDRS(functionalassessment).
Score0.
70.
21.
50.
063TrailBLogs0.
130.
060.
320.
081UHDRSindependencescaleScore1.
12.
90.
60.
100Weightkg0.
60.
82.
10.
190VerbalFluencyLogvalue0.
040.
130.
060.
221MMSEScore0.
10.
30.
50.
263OTSproblemssolvedonfirstchoiceLogodds0.
060.
180.
310.
300SystolicbloodpressuremmHg0.
955.
093.
160.
318TrailALogs0.
010.
110.
130.
429PreEDerrorsLogodds0.
050.
700.
780.
4552462JNeurol(2017)264:2457–2463metabolitesthathadpreviouslybeenassociatedwithpro-gressionofHD[16].
Thesecomplexmultivariateresultswillbereportedinaseparatepaper.
Haematological,biochemicalandECGchangesOftheroutinebloodsamplestakenforhaematological,renalandliverfunction,noconsistentorsustainedchangeswereseen.
Oneindividualshowedatransientriseincre-atinekinasewhichsettledbytheirnextvisit.
Twoindividu-alsshowedmildandtemporaryderangementoftheirLFTswhich,onreviewbyahepatologist,requirednofurtherinvestigationortreatment.
AllECGswerereviewedbyaconsultantcardiologist.
TwoECGsshowedventricularectopicsduringthestudy.
Follow-upshowednounderlyingrhythmabnormalityandnotreatmentwasrequiredduringorafterthestudy.
OtherAEsaresummarisedinsupplementaryTable2.
DiscussionThisstudyhasshownthatitisfeasibletoundertakeatrialofrilmenidineinpatientswithmildormoderateHDandthatrilmenidinewaswelltolerated.
Whilethis2-yearopen-labelstudyreportednodrug-relatedseriousadverseeventsandwithdrawals,itwasalsonotabletoascertainwhethertherewasanyefficacyofthisagentasthetrialwasnotdesignedtotestfordrug-relatedchangesinsecondaryoutcomemeasures.
However,thedatadoshowchangesthatarelessthan,orequal,tothatexpectedinpatientsatthisstageofdiseaseusingrecentlargehistoricalcontroldata[8,12].
ThisobservationneedstobeviewedcautiouslygivenwehadnoplaceboarminthisstudyandbecausethelargecohortstudiesofHDpopulationsthathavebeenreporteddifferinthewaytheyweredesignedandanalysed.
However,wedid,forexample,findalowerrateofgeneralisedbrainatrophy(1.
5%peryearseenhereversus2.
05%pointsperyear)andbasalgangliavolumeloss(+0.
8to1.
0%peryearseenhereversus7.
46%pointsperyear)thanthatseeninTRACK-HD[12],asmallerdeclineinMMSEscores(+0.
1pointsperyearseenhereversus0.
7pointsperyear)[19]andasmallerreductionintheUHDRStotalfunctionalcapacityscore(+0.
4pointsperyearseenhereversus+0.
6pointsperyear)thanintheCOHORTstudy[20].
Incon-trast,increasesinUHDRStotalmotorscoresweresimilar(+3.
5pointsperyearversus+3pointsperyear)tothatseeninboththeTRACK-HD([12]andCOHORTstudies[8]).
Weightandcognitivetestsdidnotdeclinesignificantlyoverthecourseofthestudy,thoughitisimpossibletoruleoutthepossibilityofalearningeffectwiththesecognitivetests.
Wealsoexaminedmetabolomicchangesovertime,givenwehavepreviouslyfounddifferencesinpatientswithHDandcontrols[16].
Wefoundnodifferencesoverthecourseofthestudy.
ThisisthefirstlongitudinalstudyinHDtoinves-tigatemetabolomicanalysisasapossiblestudyendpoint.
Thisstudyhasanumberoflimitations.
First,itisopenlabelwithasmallnumberofpatients.
Second,therewereseveraldropoutstowardstheendofthetrial,whichalthoughnotdrug-related,neverthelessreducedthepowertodrawanyconclusions.
Third,thestudyfollowedpatientsforonly2yearsbuteffectsondiseasemodification,ineitherdirec-tion,mayneedlongerfollow-uptobecomeapparent.
Finally,wewereunabletomeasuretargetengagementintheCNSintermsofwhethertherilmenidinetrulydidup-regulateautophagyattheintendedsite.
Untilthiscanberesolved,studiesofthistypecanonlypostulatethatanyeffectsaremediatedviathisintracellularpathway.
Table4ChangesinregionalbrainvolumeonMRI(orderedbythesignificanceofthepvalue)Percentvolumechangeperyearforselectedregionspvaluesfromone-samplettestOnlyoneregionhadasignificantlynon-normaldistributionaccord-ingtoShapiro–Wilkwhichwasrighthippocampus(p=0.
04)RegionPercentchangepvalueLeftlateralventricle7.
2<0.
001Rightlateralventricle6.
6<0.
001Brainvolume1.
30.
001Brainvolumeexc.
ventricle1.
80.
001Leftputamen4.
40.
002Rightinferiorlateralventricle8.
10.
002Brainstem0.
90.
003Leftcerebellumcortex0.
90.
003Pons0.
80.
003Leftinferiorlateralventricle9.
60.
005Rightcerebellumcortex1.
00.
009Totalgreymattervolume1.
50.
016Cerebralwhitemattervolume2.
20.
019Subcortgreymattervolume1.
20.
02Cerebralcortexvolume1.
80.
038Midbrain0.
40.
174Rightthalamusproper1.
40.
195Rightputamen2.
10.
282Leftamygdala0.
70.
296Leftthalamusproper0.
90.
328Rightamygdala0.
60.
487Medulla0.
40.
549Righthippocampus0.
20.
618Leftcaudate0.
80.
621Leftaccumbensarea1.
20.
75Rightcaudate1.
00.
797Rightaccumbensarea0.
40.
905Lefthippocampus0.
00.
9572463JNeurol(2017)264:2457–2463Insummary,thisstudyhasshownthatrilmenidineappearstoberelativelysafeandwelltoleratedinclini-callymanifestHuntington'sdisease.
Whetheritslowsdowndiseaseprogressionthroughaneffectonautophagyisunresolved,butthedatafromourtrialwouldencour-ageundertakingfurtherstudieswiththisagentinlarger,randomised,placebo-controlledtrials.
CompliancewithethicalstandardsFundingThisworkwassupportedbyanNIHRgrantofaBiomedi-calResearchCentretotheUniversityofCambridge/AddenbrookesHospital,WellcomeTrust(PrincipalResearchFellowshiptoDCR(095317/Z/11/Z)andJBR(SeniorResearchFellowship,103838)andDCRisgratefulforfundingfromtheUKDementiaResearchInstitute(fundedbytheMRC,Alzheimer'sResearchUKandtheAlzheimer'sSociety).
InadditiontheimagingpartofthestudywassupportedbyagrantfromtheRosetreesTrust.
ConflictsofinterestNoneoftheauthorsreportedanyconflictsofinterest.
EthicalstandardsAllhumanstudieshavebeenapprovedbytheappropriateethicscommitteeandhavethereforebeenperformedinaccordancewiththeethicalstandardslaiddowninthe1964Declara-tionofHelsinki.
OpenAccessThisarticleisdistributedunderthetermsoftheCreativeCommonsAttribution4.
0InternationalLicense(http://crea-tivecommons.
org/licenses/by/4.
0/),whichpermitsunrestricteduse,distribution,andreproductioninanymedium,providedyougiveappro-priatecredittotheoriginalauthor(s)andthesource,providealinktotheCreativeCommonslicense,andindicateifchangesweremade.
References1.
BatesGP,TabriziSJ,JonesL(2014)Huntington'sdisease,Oxfordmonographsinmedicalgenetics,4thedn.
OxfordUni-versityPress,Oxford2.
MasonSL,BarkerRA(2016)AdvancingpharmacotherapyfortreatingHuntington'sdisease:areviewoftheexistingliterature.
ExpertOpinPharmacother17(1):41–523.
AndrewSEetal(1993)Therelationshipbetweentrinucleotide(CAG)repeatlengthandclinicalfeaturesofHuntington'sdis-ease.
NatGenet4(4):398–4034.
RavikumarB,RubinszteinDC(2006)Roleofautophagyintheclearanceofmutanthuntingtin:asteptowardstherapyMolAspectsMed27(5–6):520–5275.
BergerZetal(2006)Rapamycinalleviatestoxicityofdifferentaggregate-proneproteins.
HumMolGenet15(3):433–4426.
RennaMetal(2010)Chemicalinducersofautophagythatenhancetheclearanceofmutantproteinsinneurodegenerativediseases.
JBiolChem285(15):11061–110677.
RoseCetal(2010)Rilmenidineattenuatestoxicityofpolyglu-tamineexpansionsinamousemodelofHuntington'sdisease.
HumMolGenet19(11):2144–21538.
HuntingtonStudyGroup(2012)C.
I.
andE.
Dorsey,Characteri-zationofalargegroupofindividualswithhuntingtondiseaseandtheirrelativesenrolledintheCOHORTstudy.
PLoSOne7(2):e295229.
TabriziSJetal(2011)Biologicalandclinicalchangesinpremani-festandearlystageHuntington'sdiseaseintheTRACK-HDstudy:the12-monthlongitudinalanalysis.
LancetNeurol10(1):31–4210.
BechtelNetal(2010)TappinglinkedtofunctionandstructureinpremanifestandsymptomaticHuntingtondisease.
Neurology75(24):2150–216011.
TabriziSJetal(2012)PotentialendpointsforclinicaltrialsinpremanifestandearlyHuntington'sdiseaseintheTRACK-HDstudy:analysisof24monthobservationaldata.
LancetNeurol11(1):42–5312.
TabriziSJetal(2013)Predictorsofphenotypicprogressionanddiseaseonsetinpremanifestandearly-stageHuntington'sdiseaseintheTRACK-HDstudy:analysisof36-monthobservationaldata.
LancetNeurol12(7):637–64913.
ShoulsonI,FahnS(1979)Huntingtondisease:clinicalcareandevaluation.
Neurology29(1):1–314.
KukullWAetal(1994)TheMini-MentalStateExaminationscoreandtheclinicaldiagnosisofdementia.
JClinEpidemiol47(9):1061–106715.
RobbinsTWetal(1994)CambridgeNeuropsychologicalTestAutomatedBattery(CANTAB):afactoranalyticstudyofalargesampleofnormalelderlyvolunteers.
Dementia5(5):266–28116.
UnderwoodBRetal(2006)Huntingtondiseasepatientsandtrans-genicmicehavesimilarpro-catabolicserummetaboliteprofiles.
Brain129:877–88617.
StanDevelopmentTeam(2016)RStan:theRinterfacetoStan,Version2.
9.
0.
http://mc-stan.
org/18.
StanDevelopmentTeam(2016)Stanmodelinglanguageusersguideandreferencemanual,version2.
9.
0.
http://mc-stan.
org19.
DorseyERetal(2013)Naturalhistoryofhuntingtondisease.
JAMANeurol70:1520–153020.
MarderKetal(2000)RateoffunctionaldeclineinHuntington'sdisease.
HuntingtonStudyGroup.
Neurology54(2):452–458
1007/s00415-017-8647-0ORIGINALCOMMUNICATIONAnopenlabelstudytoassessthefeasibilityandtolerabilityofrilmenidineforthetreatmentofHuntington'sdiseaseBenjaminR.
Underwood1·ZeynW.
GreenThompson2·PeterJ.
Pugh2·StanleyE.
Lazic3·SarahL.
Mason7·JulesGriffin4·P.
SimonJones5·JamesB.
Rowe5·DavidC.
Rubinsztein6,7·RogerA.
Barker2,5,8Received:11September2017/Revised:9October2017/Accepted:11October2017/Publishedonline:26October2017TheAuthor(s)2017.
Thisarticleisanopenaccesspublicationindisease-specificvariables,suchasmotor,cognitiveandfunctionalperformance,structuralMRIandserummetabo-lomicanalysis.
12patientscompletedthestudy;reasonsforwithdrawalincludedproblemstoleratingstudyprocedures(MRI,andvenepuncture),depressionrequiringhospitaladmissionandlogisticalreasons.
Threeseriousadverseeventswererecorded,includinghospitalisationfordepres-sion,butnonewerethoughttobedrug-related.
Changesinsecondaryoutcomeswereanalysedastheannualrateofchangeinthestudygroup.
Theoverallchangewascom-parabletochangesseeninrecentlargeobservationalstud-iesinHDpatients,thoughdirectstatisticalcomparisonstothesestudieswerenotmade.
Chronicoraladministrationofrilmenidineisfeasibleandwell-toleratedandfuture,larger,placebo-controlled,studiesinHDarewarranted.
Trialregistration:EudraCTnumber2009-018119-14.
KeywordsAutophagy·Neurodegenerativedisease·Interventionaltrial·MRI·CognitionIntroductionHuntington'sdisease(HD)isaneurodegenerativedisorderwhichpresentswithacombinationofmovement,psychi-atricandcognitivedeficits(reviewedinBatesetal.
[1]).
Ittypicallyprogresseswithincreasingdisabilitytodeathoverthecourseof15–20years.
Therearecurrentlynodisease-modifyingtreatmentsavailablewhichcanaltertheprogres-sionofHD[2].
Fromtheveryfirstdescription,itwasrecognisedthattheconditionisfamilial,andtheunderlyinggeneticmutation,anexpandedtrinucleotiderepeatintheHuntingtingeneontheshortarmofchromosomefour,wasidentifiedin1993[3].
ThishasledtoabetterunderstandingofthemolecularAbstractPreclinicaldatahaveshownthatrilmenidinecanregulateautophagyinmodelsofHuntington'sdisease(HD),providingapotentialroutetoalterthediseasecourseinpatients.
Consequently,a2-yearopen-labelstudyexam-iningthetolerabilityandfeasibilityofrilmenidineinmild-moderateHDwasundertaken.
18non-dementedpatientswithmildtomoderateHDtookdailydosesof1mgRilme-nidinefor6monthsand2mgforafurther18monthsfol-lowedbya3-monthwashoutperiod.
Theprimaryoutcomewasthenumberofwithdrawalsandseriousadverseevents.
SecondaryoutcomesincludedsafetyparametersandchangesElectronicsupplementarymaterialTheonlineversionofthisarticle(doi:10.
1007/s00415-017-8647-0)containssupplementarymaterial,whichisavailabletoauthorizedusers.
*RogerA.
Barkerrab46@cam.
ac.
uk1FulbournHospital,Fulbourn,CambridgeCB215EF,UK2Addenbrooke'sHospital,HillsRoad,CambridgeCB212QQ,UK3QuantitativeBiology,DiscoverySciences,IMEDBiotechUnit,AstraZeneca,CambridgeCB40WG,UK4DepartmentofBiochemistry,UniversityofCambridge,CambridgeCB21GA,UK5DepartmentofClinicalNeurosciences,andMRCCognitionandBrainSciencesUnit,UniversityofCambridge,CambridgeBiomedicalCampus,CambridgeCB20SZ,UK6CambridgeInstituteforMedicalResearch,UniversityofCambridge,CambridgeBiomedicalCampus,HillsRoad,CambridgeCB20XY,UK7UKDementiaResearchInstitute,UniversityofCambridge,HillsRoad,CambridgeCB20XY,UK8JohnVanGeestCentreforBrainRepair,ForvieSite,RobinsonWay,CambridgeCB20PY,UK2458JNeurol(2017)264:2457–2463pathophysiologyofthecondition,andconsequentlytorationaltreatmentapproaches.
Oneexampleofthishasbeentherecog-nitionthatthemutantproteinproducedbythegeneticmutationisasubstratefordegradationbyautophagy[4].
Autophagydescribesaprocesswhereintracytoplasmicves-icles,calledautophagosomes,delivercytoplasmiccontentstolysosomesfordegradation.
Itisamenabletoup-regulationbydrugsinavarietyofcellular,fly,zebrafishandmousemodelsofHD,andthisstrategyamelioratessignsofthediseaseinthesemodels.
Theoriginaldrugthatwasshowntodothiswasrapamycin,aninhibitorofthemammaliantargetofrapamycinormTOR[5].
However,whilethisdrugisapprovedforhumanuse,ithasasignificantsideeffectprofileincludingimmu-nosuppressionmakingitanunattractiveoptionfortriallinginpatientswithchronicneurodegenerativedisordersoftheCNS.
Subsequentdrugscreenshaveidentifiedlesstoxicup-regulatorsofautophagywhichstillretaintheabilitytorescuecellsandbehaviourinmammalianandothermodelsofHD[6].
Onesuchdrugisrilmenidine(N-(dicyclopropylmethyl)-4,5-dihydro-2-oxazolamine),anα2receptorantagonistandanimidazolineI1receptoragonist[7].
Ithasbeenextensivelyusedintheclinicasacentrallyactinganti-hypertensiveagentwithnosignificantsideeffectscomparedtoplaceboatdosesof1mgperdayandwithanextensivesafetyrecordinhumanuseincludingelderlysubjects.
Thus,thisagentisanidealcandidatetoassessforthefeasibilityandtolerabilityofthisapproachinpatientswithHD.
Investigatingwhethersuchtreatmentscanreallyslowdiseaseprogressioninchronicneurodegenerativedisorders,suchasHD,willrequirelargeexpensivestudies.
Indeed,longitudinalobservationalstudiesofpatientswithHunting-ton'sdiseaseover2–3years(forexample,COHORT[8]andTRACK-HD[9–12])havehighlightedthatdefinitivelystudyingtheeffectsofdisease-modifyingagentsonHDwillrequirelargenumbersofpatientsfollowedforseveralyears.
However,beforeembarkingonstudiesofthistype,itisimportanttoascertainthatthedrugtobetriallediswelltoleratedandthestudyfeasible.
WethereforeundertookafirstinHDsmallopen-labelstudyovera2-yearperiodlookingatthetolerabilityandfeasibilityofrilmenidineasapossibledisease-modifyingagent.
Whilenoaccountofitseffectivenesscanbecon-cludedfromsuchastudy,ithasprovidedencouragingdatawhichcannowbeusedtoplanmoredefinitivelarge-scale,multi-centre,double-blind,placebocontroltrials.
MethodsTrialdesignandparticipantsThiswasasinglecentreopen-labelstudycarriedoutwithpatientsrecruitedfromtheregionalHDclinicattheCambridgeUniversityHospitalsNHSTrust(Addenbrooke'sHospital),UK.
Writteninformedconsentwasobtainedfromallparticipants.
Inclusioncriteria1.
AconfirmeddiagnosisofHuntington'sdiseaseonthebasisofqualifyingclinicalsignsandsymptoms,spe-cificallyaUnifiedHuntington'sDiseaseRatingScale1999(UHDRS)totalmotorscoreofatleast5[9],andconfirmationofaCAGrepeatof>36inexon1ofthehttgene.
2.
HDstage2–3asdefinedusingaUHDRStotalfunc-tionalcapacity(TFC)scoreofgreaterthan4[13].
3.
Ambulantandabletoself-careindependently.
4.
Agedbetween18and70.
5.
Englishspeakingandabletogivewritten,informedcon-sent.
Exclusioncriteria1.
Anongoingclinicallysignificantandunstablegen-eralmedicalcondition[includingbutnotlimitedto;asthma,chronicobstructivepulmonarydisease(COPD),ongoingischaemicheartdiseaseproblems(IHD),congestivecardiacfailure(CCF),leftbundlebranchblock(LBBB)oracerebrovascularaccident(CVA)]confirmedviapastmedicalhistoryorbaselinemedicalorphysicalexaminationandinvestigations.
2.
Prescribedanti-hypertensivemedicationoranydrugknowntobecontraindicatedortohaveanadverseinteractionwithrilmenidine(viz.
amonoamineoxi-daseinhibitor).
3.
Knownhypersensitivitytorilmenidine.
4.
Ongoingsignificantmentalillnessdeterminedbyevi-dence,orahistory,ofapsychoticoraffective(depres-sionormania)episodeinthe6monthspriortobase-lineInterviewasassessedusingtheDiagnosticandStatisticalManualofMentalDisorderscriteria(FourthEditionwithTextRevision;AmericanPsychiatricAssociation).
5.
Prescribedtypicaloratypicalanti-psychoticmedica-tionwhenbeingexplicitlyusedtotreatapsychoticillness(asopposedtothemovementdisorderofHD).
6.
Pregnantorbreastfeedingfemalepatients,includingthoseplanningtoconceiveduringtheperiodofthetrial.
Womenofchildbearingagewhowereneitherpregnantnorplanningtoconceiveduringtheperiodofthestudyweredeemedeligibleprovidedtheyusedtwoformsofcontraception,atleastoneofwhichhadtobeabarriermethod.
2459JNeurol(2017)264:2457–24637.
Substance(alcoholorillegal/prescriptiondrug)misuseinthe6monthspriortothebaselineassessment.
8.
Knownco-morbidmajorneurologicaldisorder(includ-ingParkinson'sdiseaseoranestablisheddementia),HIV/AIDSorhepatitis(HBVorHCV).
9.
Previousneurosurgerytothebrain.
10.
Markedclinicallyadverseabnormalitiesonlaboratoryinvestigationsincludingcreatinineclearance177Umol/l.
InterventionsandoutcomesAllpatientsweregiven1mgrilmenidinedailyfor6monthsandthen2mgdailyforthenext18monthsofthestudy.
Followingscreening,patientswereseenforabaselineassessmentandthenreviewedatmonths1,3,6,9,12,18,24and27(i.
e.
3monthsafterstop-pingtherilmenidine).
Seriousadverseevents(SAEs)andwithdrawals(allprimaryendpoints)wererecordedalongwithnon-seriousadverseevents,height,weight,vitalsigns,routinehaematologyandbiochemistrybloodmeasuresandECGoccurredasperthescheduleofevents(Table1).
SpecificmeasuresoftheprogressionoftheirHDincludedUHDRStotalmotorscore,functionalandindependencescales[13]aswellastheirperformanceonthetrailmak-ingtest,MiniMentalStateExamination(MMSE)[14]andverbalfluencytasks(asassessedbythecontrolledoralwordassociationtest,COWAT).
OthercognitivetestsfromtheCambridgeAutomatedNeuropsychologicalTestAutomatedBattery(CANTAB)[15]werealsousedandincludedthemeantimetaken(latency)andtotalnumberofcorrectlysolvedproblemsontheOneTouchStockingsofCambridge,andthetotalnum-beroferrorsmadepriortoandattheextra-dimensionalshiftstageoftheCANTABintra-dimensional/extra-dimensionalset-shiftingtask(ID/ED)[15]).
ThescheduleofeventsisshowninTable1.
SamplesizeandstatisticalmethodsTheestimateoftherequiredsamplesize(n=16)wasdesignedtodetectSAEs/AEsratherthanasignificantchangeinsecondaryendpointsandwasbasedondatagatheredfromourpreviousstudiesusingmetabolomicbiomarkersinHDpatients[16].
Thenumberofseriousadverseeventsanddropoutswasanalysedwithanexactbinomialtest,testingwhethertheratesweregreaterthananacceptablesafetylevelof5%peryear,or10%overthecourseofthestudy.
One-sidedpvaluesand95%CIarereported.
Forsecondaryoutcomes,themeanrateofprogression(slope)wasthemainparameterofinterest.
Theseoutcomeswereanalysedwithmultilevelmodelsallowingforpatient-specificinterceptsandslopes.
Thebasicmodelforthesec-ondaryanalyseswaswhereyijistheoutcomeforpatientiattimej,andαpatientandβpatientarethepatient-specificinterceptsandslopes,respec-tively.
Priorsformodelparameterswerenon-informative(oryijNormal(patient[i]+patient[i]timeij,),Table1AssessmentscheduleforparticipantsinthetrialBaselineOnemonthreviewMonths3,6,9,12,18and2427monthsMedicalhistory,familyhistoryandphysicalexaminationXXxXWeightXXxXVitalsigns(bloodpressureandpulse/heartrate)XXxXHaematologyXXxXBiochemistryXXxX12-LeadECGXXxXReviewofadverseeventsXXxXBloodsampleformetabolomicsXxXReviewofconcomitantmedicationsXxXMagneticresonanceimaging(brain)XXUnifiedHuntington'sDiseaseRatingScaleXxXMinimentalStateExaminationXxXNeuropsychiatricInventoryXxxTrailmakingtestXxXVerbalfluencyXxXTowerofLondon(OneTouchStockingsofCambridge)XxXExtra-dimensional/intra-dimensionalshiftsXxX2460JNeurol(2017)264:2457–2463verymildlyinformativetoaidconvergence).
Forpositivelyskewedoutcomes,thedatawerelog-transformedandsomeoutcomesusedabinomialornegativebinomiallikelihoodifthedatawasbettermodelledasproportionsorcounts.
Theresultsarepresentedastheannualrateofchangeandtheconfidenceintervalsarethe95%highestposteriordensityintervals.
pvaluesrepresenttheproportionoftheposte-riordensityontheoppositesideofzerofromtheestimatedmean.
Forexample,theUHDRSmotorscoreincreasedby3.
5unitsannually,andtheassociatedpvalueof0.
0063tellsusthat0.
63%oftheposteriordistributionhadnegativevalues.
ThedatawereanalysedwiththeBayesiansoftware"Stan"andtherstanandrethinkingRpackages[17,18].
Giventhelargenumberofsecondaryvariablesandthelackofdirecthypothesistesting,nostatisticaladjustmenthasbeenmadeformultipletesting.
Imaging16patientsunderwentstructuralMRimagingattheCambridgeBiomedicalCampususinga1.
5TGEMedi-calSystemsDiscoveryMR450scanner.
StructuralMRIscanswerecollectedpriortocommencementoftreatmentandat27months(i.
e.
3monthsaftertreatmentceased)toavoidanyeffectsofthemedicationonbrainvolumesandfluidshifts,givenitsknownanti-hypertensiveactions.
AT1-weighted3DBravofastspoiledgradientecho(SPGR)imagewasacquiredwithrepetitiontime(TR)=8156ms,echotime(TE)=3.
18ms,matrix=256*256,in-planeresolutionof1*1mm,252slicesof1mmthickness,inver-siontime=900msandflipangle=9°.
AnalysisoftheMRIdatawasperformedusingFreesurferv.
6(stable-v6-beta-20151015).
Theregionofinterest(ROI)valueswasimportedintoSPSS.
Apairedttestwasusedtoestimatethechangesinvolumebetweenthetwotime-points.
BloodsamplingandmetabolomicanalysisBloodsamplingformetabolomicanalysisbygaschromatog-raphy–massspectrometrywasalsoundertakenusingtech-niquespreviouslydescribed[16].
Metabolomicanalysisisatechniquewhichprovidesmultivariatequantitativeanalysisofmolecularfragments.
Measuringthepresenceofalargenumberofthesefragmentsandtheirrelativeabundanceleadstoamolecularprofilewhichcanhelpdifferentiatebetweendiseasestatesandmayalsobeusefulasapotentialbiomarkerofprogression.
ThereisreasontobelievethatmetabolismisalteredinHDandanumberofpapershavenowdescribedmetabolomicchangesinHD,includingfromourowngroup[16].
Inthisstudy,weperformedsequen-tiallongitudinalmetabolomicanalysisineachpatientasanexploratorypotentialbiomarkerofdiseaseprogression.
ResultsEighteenpatientswererecruitedtothestudy.
OnedroppedoutbeforethetreatmentwasstartedasthepatientwasunabletotolerateMRIscanning.
Afurtherindividualdroppedoutatthe1-monthvisitasthepatientwasunabletotoleratevenepuncture.
Thisleft16patientsforwhomthereweremorethanoneobservationandwhoformedthetrialpopulation.
Onepatientwithdrewfromthestudyafter6monthswhentheybecamedepressedandrequiredpsychiatricadmission.
Onefurtherpatientdroppedoutat21monthsasthepatientwasnolongerabletotraveltoclinicandtwoafterthe24-monthvisit,onewhonolongerwishedtocontinueinthetrialandtheotherwaslosttofol-low-up.
Thus,12patientscompletedthe27-monthfollow-upvisit.
ExcessivemovementartefactsintheT1imageofthefollow-upvisitofoneparticipantmeantthat11patientswereincludedinthelongitudinalimagingstudy.
ThisissummarisedinFig.
1.
Screenedn=18Excluded-unabletotolerateMRIn=1Receivedintervenonn=17Withdrewn=1Completed6monthfollow-upn=16Withdrewn=2Completed24monthfollow-upn=14Fig.
1Patientflowthroughthestudy2461JNeurol(2017)264:2457–2463RecruitmentRecruitmentoccurredbetweenOctober2012andJuly2013.
Thefinalpatientfinalvisit(FPFV)wascompletedinJune2015.
BaselinedataBaselinedemographicandclinicalcharacteristicsareshowninTable2.
PrimaryoutcomemeasuresOfthe18patientswhowererecruitedtothestudy,6with-drewforthereasonsgivenabove.
Thenumberofwith-drawalswasgreaterthanthepre-specifiedtargetrateof0.
1(p=0.
0002,6/18patients=0.
33dropoutrate;95%CI0.
16–1.
0).
However,noneofthewithdrawalswereconsid-eredtobedrug-related.
Threeseriousadverseevents(SAEs)werereported:onebrokenwristfromaphysicalfall,oneadmissiontohospitalformigraineaftertheindividualwithdrewtheirmigraineprophylaxistreatment,andoneadmissionfordepressionfol-lowingcessationofantidepressants.
Nonewerethoughttobedrug-related.
SecondaryoutcomemeasuresThesecondaryoutcomemeasuresaresummarisedinTable3(secondaryoutcomemeasures)andTable4(changesinregionalbrainvolumeonMRI).
Ofthesec-ondaryendpoints,theUHDRStotalmotorscore(+3.
5,95%CI0.
8–6.
3,p=0.
009),wholebrainvolume(1.
3%,p=0.
001)andUHDRSfunctionalcapacity(0.
4,95%CI0.
8–0.
02,p=0.
020)showedadeclineoverthestudyperiodasexpected.
Intermsofcognitivetests,latencytocorrecttimeontheCANTABOneTouchStockingsofCambridgeimproved(0.
12,95%CI0.
23to0.
01,p=0.
014),withnosig-nificantchangesinanyoftheothercognitivetests.
TheMRIdatarevealedadeclineintotalbrainvolume(excludingventricles)of1.
8%peryear(p=0.
001),althoughwithrespecttothebasalganglia,onlytheleftputa-menshowedasignificantdecreaseinsizeoverthecourseofthestudy.
Othersecondaryendpointsdidnotsignificantlychangeoverthestudy.
AllotherendpointsremainedunchangedincludingthemetabolomicanalysiswherenosignificantdifferenceswerenotedbetweenbaselineandtheendofstudyincludinginTable2BaselinedemographicandclinicalcharacteristicsBMIbodymassindex,MMSEMiniMentalStateExam,UHDRSUni-fiedHuntington'sDiseaseRatingScaleSex11male5femaleAgeMean53(range37–69)Weight80.
2kg(62.
5–100)BMI27.
5(21.
4–33.
4)MMSE27.
4(24–30)Verbalfluency23.
2(6–53)UHDRSmotor24.
5(7–46)UHDRSfunctionalcapacity9.
62(5–13)UHDRSfunctionalassessment28.
2(25–35)UHDRSindependence80(70–100)Table3Secondaryoutcomemeasures(orderedbysignificanceofthepvalue)CANTABOneTouchStockingsofCambridgeEDextra-dimensionalshiftontheCANTABintra-dimensional/extra-dimensionalset-shiftingtask,MMSEMiniMentalStateExamination,UHDRSUnifiedHuntington'sDiseaseRatingScaleVariableUnitsChangeperannumLowerCIUpperCIpvalueUHDRS(motor)Score3.
50.
86.
30.
009OTS-latencytocorrectLogs0.
120.
230.
010.
014UHDRS(totalfunctionalcapacity)Score0.
40.
80.
020.
020EDSerrorsNumber0.
20.
50.
060.
050UHDRS(functionalassessment).
Score0.
70.
21.
50.
063TrailBLogs0.
130.
060.
320.
081UHDRSindependencescaleScore1.
12.
90.
60.
100Weightkg0.
60.
82.
10.
190VerbalFluencyLogvalue0.
040.
130.
060.
221MMSEScore0.
10.
30.
50.
263OTSproblemssolvedonfirstchoiceLogodds0.
060.
180.
310.
300SystolicbloodpressuremmHg0.
955.
093.
160.
318TrailALogs0.
010.
110.
130.
429PreEDerrorsLogodds0.
050.
700.
780.
4552462JNeurol(2017)264:2457–2463metabolitesthathadpreviouslybeenassociatedwithpro-gressionofHD[16].
Thesecomplexmultivariateresultswillbereportedinaseparatepaper.
Haematological,biochemicalandECGchangesOftheroutinebloodsamplestakenforhaematological,renalandliverfunction,noconsistentorsustainedchangeswereseen.
Oneindividualshowedatransientriseincre-atinekinasewhichsettledbytheirnextvisit.
Twoindividu-alsshowedmildandtemporaryderangementoftheirLFTswhich,onreviewbyahepatologist,requirednofurtherinvestigationortreatment.
AllECGswerereviewedbyaconsultantcardiologist.
TwoECGsshowedventricularectopicsduringthestudy.
Follow-upshowednounderlyingrhythmabnormalityandnotreatmentwasrequiredduringorafterthestudy.
OtherAEsaresummarisedinsupplementaryTable2.
DiscussionThisstudyhasshownthatitisfeasibletoundertakeatrialofrilmenidineinpatientswithmildormoderateHDandthatrilmenidinewaswelltolerated.
Whilethis2-yearopen-labelstudyreportednodrug-relatedseriousadverseeventsandwithdrawals,itwasalsonotabletoascertainwhethertherewasanyefficacyofthisagentasthetrialwasnotdesignedtotestfordrug-relatedchangesinsecondaryoutcomemeasures.
However,thedatadoshowchangesthatarelessthan,orequal,tothatexpectedinpatientsatthisstageofdiseaseusingrecentlargehistoricalcontroldata[8,12].
ThisobservationneedstobeviewedcautiouslygivenwehadnoplaceboarminthisstudyandbecausethelargecohortstudiesofHDpopulationsthathavebeenreporteddifferinthewaytheyweredesignedandanalysed.
However,wedid,forexample,findalowerrateofgeneralisedbrainatrophy(1.
5%peryearseenhereversus2.
05%pointsperyear)andbasalgangliavolumeloss(+0.
8to1.
0%peryearseenhereversus7.
46%pointsperyear)thanthatseeninTRACK-HD[12],asmallerdeclineinMMSEscores(+0.
1pointsperyearseenhereversus0.
7pointsperyear)[19]andasmallerreductionintheUHDRStotalfunctionalcapacityscore(+0.
4pointsperyearseenhereversus+0.
6pointsperyear)thanintheCOHORTstudy[20].
Incon-trast,increasesinUHDRStotalmotorscoresweresimilar(+3.
5pointsperyearversus+3pointsperyear)tothatseeninboththeTRACK-HD([12]andCOHORTstudies[8]).
Weightandcognitivetestsdidnotdeclinesignificantlyoverthecourseofthestudy,thoughitisimpossibletoruleoutthepossibilityofalearningeffectwiththesecognitivetests.
Wealsoexaminedmetabolomicchangesovertime,givenwehavepreviouslyfounddifferencesinpatientswithHDandcontrols[16].
Wefoundnodifferencesoverthecourseofthestudy.
ThisisthefirstlongitudinalstudyinHDtoinves-tigatemetabolomicanalysisasapossiblestudyendpoint.
Thisstudyhasanumberoflimitations.
First,itisopenlabelwithasmallnumberofpatients.
Second,therewereseveraldropoutstowardstheendofthetrial,whichalthoughnotdrug-related,neverthelessreducedthepowertodrawanyconclusions.
Third,thestudyfollowedpatientsforonly2yearsbuteffectsondiseasemodification,ineitherdirec-tion,mayneedlongerfollow-uptobecomeapparent.
Finally,wewereunabletomeasuretargetengagementintheCNSintermsofwhethertherilmenidinetrulydidup-regulateautophagyattheintendedsite.
Untilthiscanberesolved,studiesofthistypecanonlypostulatethatanyeffectsaremediatedviathisintracellularpathway.
Table4ChangesinregionalbrainvolumeonMRI(orderedbythesignificanceofthepvalue)Percentvolumechangeperyearforselectedregionspvaluesfromone-samplettestOnlyoneregionhadasignificantlynon-normaldistributionaccord-ingtoShapiro–Wilkwhichwasrighthippocampus(p=0.
04)RegionPercentchangepvalueLeftlateralventricle7.
2<0.
001Rightlateralventricle6.
6<0.
001Brainvolume1.
30.
001Brainvolumeexc.
ventricle1.
80.
001Leftputamen4.
40.
002Rightinferiorlateralventricle8.
10.
002Brainstem0.
90.
003Leftcerebellumcortex0.
90.
003Pons0.
80.
003Leftinferiorlateralventricle9.
60.
005Rightcerebellumcortex1.
00.
009Totalgreymattervolume1.
50.
016Cerebralwhitemattervolume2.
20.
019Subcortgreymattervolume1.
20.
02Cerebralcortexvolume1.
80.
038Midbrain0.
40.
174Rightthalamusproper1.
40.
195Rightputamen2.
10.
282Leftamygdala0.
70.
296Leftthalamusproper0.
90.
328Rightamygdala0.
60.
487Medulla0.
40.
549Righthippocampus0.
20.
618Leftcaudate0.
80.
621Leftaccumbensarea1.
20.
75Rightcaudate1.
00.
797Rightaccumbensarea0.
40.
905Lefthippocampus0.
00.
9572463JNeurol(2017)264:2457–2463Insummary,thisstudyhasshownthatrilmenidineappearstoberelativelysafeandwelltoleratedinclini-callymanifestHuntington'sdisease.
Whetheritslowsdowndiseaseprogressionthroughaneffectonautophagyisunresolved,butthedatafromourtrialwouldencour-ageundertakingfurtherstudieswiththisagentinlarger,randomised,placebo-controlledtrials.
CompliancewithethicalstandardsFundingThisworkwassupportedbyanNIHRgrantofaBiomedi-calResearchCentretotheUniversityofCambridge/AddenbrookesHospital,WellcomeTrust(PrincipalResearchFellowshiptoDCR(095317/Z/11/Z)andJBR(SeniorResearchFellowship,103838)andDCRisgratefulforfundingfromtheUKDementiaResearchInstitute(fundedbytheMRC,Alzheimer'sResearchUKandtheAlzheimer'sSociety).
InadditiontheimagingpartofthestudywassupportedbyagrantfromtheRosetreesTrust.
ConflictsofinterestNoneoftheauthorsreportedanyconflictsofinterest.
EthicalstandardsAllhumanstudieshavebeenapprovedbytheappropriateethicscommitteeandhavethereforebeenperformedinaccordancewiththeethicalstandardslaiddowninthe1964Declara-tionofHelsinki.
OpenAccessThisarticleisdistributedunderthetermsoftheCreativeCommonsAttribution4.
0InternationalLicense(http://crea-tivecommons.
org/licenses/by/4.
0/),whichpermitsunrestricteduse,distribution,andreproductioninanymedium,providedyougiveappro-priatecredittotheoriginalauthor(s)andthesource,providealinktotheCreativeCommonslicense,andindicateifchangesweremade.
References1.
BatesGP,TabriziSJ,JonesL(2014)Huntington'sdisease,Oxfordmonographsinmedicalgenetics,4thedn.
OxfordUni-versityPress,Oxford2.
MasonSL,BarkerRA(2016)AdvancingpharmacotherapyfortreatingHuntington'sdisease:areviewoftheexistingliterature.
ExpertOpinPharmacother17(1):41–523.
AndrewSEetal(1993)Therelationshipbetweentrinucleotide(CAG)repeatlengthandclinicalfeaturesofHuntington'sdis-ease.
NatGenet4(4):398–4034.
RavikumarB,RubinszteinDC(2006)Roleofautophagyintheclearanceofmutanthuntingtin:asteptowardstherapyMolAspectsMed27(5–6):520–5275.
BergerZetal(2006)Rapamycinalleviatestoxicityofdifferentaggregate-proneproteins.
HumMolGenet15(3):433–4426.
RennaMetal(2010)Chemicalinducersofautophagythatenhancetheclearanceofmutantproteinsinneurodegenerativediseases.
JBiolChem285(15):11061–110677.
RoseCetal(2010)Rilmenidineattenuatestoxicityofpolyglu-tamineexpansionsinamousemodelofHuntington'sdisease.
HumMolGenet19(11):2144–21538.
HuntingtonStudyGroup(2012)C.
I.
andE.
Dorsey,Characteri-zationofalargegroupofindividualswithhuntingtondiseaseandtheirrelativesenrolledintheCOHORTstudy.
PLoSOne7(2):e295229.
TabriziSJetal(2011)Biologicalandclinicalchangesinpremani-festandearlystageHuntington'sdiseaseintheTRACK-HDstudy:the12-monthlongitudinalanalysis.
LancetNeurol10(1):31–4210.
BechtelNetal(2010)TappinglinkedtofunctionandstructureinpremanifestandsymptomaticHuntingtondisease.
Neurology75(24):2150–216011.
TabriziSJetal(2012)PotentialendpointsforclinicaltrialsinpremanifestandearlyHuntington'sdiseaseintheTRACK-HDstudy:analysisof24monthobservationaldata.
LancetNeurol11(1):42–5312.
TabriziSJetal(2013)Predictorsofphenotypicprogressionanddiseaseonsetinpremanifestandearly-stageHuntington'sdiseaseintheTRACK-HDstudy:analysisof36-monthobservationaldata.
LancetNeurol12(7):637–64913.
ShoulsonI,FahnS(1979)Huntingtondisease:clinicalcareandevaluation.
Neurology29(1):1–314.
KukullWAetal(1994)TheMini-MentalStateExaminationscoreandtheclinicaldiagnosisofdementia.
JClinEpidemiol47(9):1061–106715.
RobbinsTWetal(1994)CambridgeNeuropsychologicalTestAutomatedBattery(CANTAB):afactoranalyticstudyofalargesampleofnormalelderlyvolunteers.
Dementia5(5):266–28116.
UnderwoodBRetal(2006)Huntingtondiseasepatientsandtrans-genicmicehavesimilarpro-catabolicserummetaboliteprofiles.
Brain129:877–88617.
StanDevelopmentTeam(2016)RStan:theRinterfacetoStan,Version2.
9.
0.
http://mc-stan.
org/18.
StanDevelopmentTeam(2016)Stanmodelinglanguageusersguideandreferencemanual,version2.
9.
0.
http://mc-stan.
org19.
DorseyERetal(2013)Naturalhistoryofhuntingtondisease.
JAMANeurol70:1520–153020.
MarderKetal(2000)RateoffunctionaldeclineinHuntington'sdisease.
HuntingtonStudyGroup.
Neurology54(2):452–458
- COPD103838.com相关文档
- Analyses103838.com
- multiplied103838.com
- choose103838.com
- ical103838.com
- 挂牌103838.com
- rc_AI232374_g_at103838.com
优林云(53元)哈尔滨电信2核2G
优林怎么样?优林好不好?优林 是一家国人VPS主机商,成立于2016年,主营国内外服务器产品。云服务器基于hyper-v和kvm虚拟架构,国内速度还不错。今天优林给我们带来促销的是国内东北地区哈尔滨云服务器!全部是独享带宽!首月5折 续费5折续费!地区CPU内存硬盘带宽价格购买哈尔滨电信2核2G50G1M53元直达链接哈尔滨电信4核4G50G1M83元直达链接哈尔滨电信8核8G50G1M131元直...
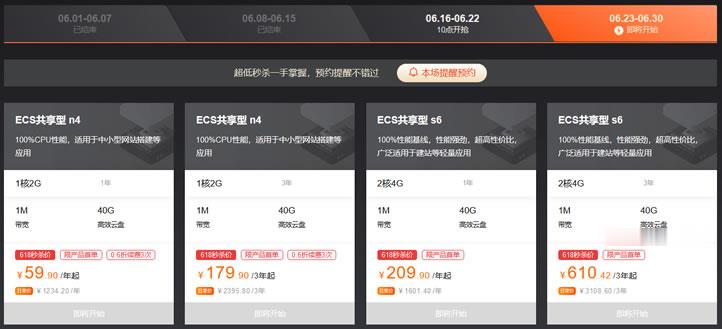
阿里云年中活动最后一周 - ECS共享型N4 2G1M年付59元
以前我们在参与到云服务商促销活动的时候周期基本是一周时间,而如今我们会看到无论是云服务商还是电商活动基本上周期都要有超过一个月,所以我们有一些网友习惯在活动结束之前看看商家是不是有最后的促销活动吸引力的,比如有看到阿里云年中活动最后一周,如果我们有需要云服务器的可以看看。在前面的文章中(阿里云新人福利选择共享性N4云服务器年79.86元且送2月数据库),(LAOZUO.ORG)有提到阿里云今年的云...
野草云99元/月 ,香港独立服务器 E3-1230v2 16G 30M 299元/月 香港云服务器 4核 8G
野草云月末准备了一些促销,主推独立服务器,也有部分云服务器,价格比较有性价比,佣金是10%循环,如果有时间请帮我们推推,感谢!公司名:LucidaCloud Limited官方网站:https://www.yecaoyun.com/香港独立服务器:CPU型号内存硬盘带宽价格购买地址E3-1230v216G240GB SSD或1TB 企盘30M299元/月点击购买E5-265016G240GB SS...

103838.com为你推荐
-
neworiental上海新东方有几个校区,分别是那几个?今日油条联通大王卡看今日头条免流量吗?mathplayerjavascript 如何判断document.body.innerHTML是否为空7788k.comwww.k6320.com 大家给我看看这网站是真是假...haokandianyingwang谁有好看电影网站啊、要无毒播放速度快的、在线等www.vtigu.com初三了,为什么考试的数学题都那么难,我最多也就135,最后一道选择,填空啊根本没法做,最后几道大题倒www.javmoo.comjavimdb怎么看baqizi.cc和空姐一起的日子电视剧在线观看 和空姐一起的日子全集在线观看bihaiyinsha以前在碧海银沙游戏城的那个打气球的游戏叫什么?干支论坛天干地支???