recommendations科讯cms
科讯cms 时间:2021-05-08 阅读:()
ClinicalQualityMeasuresforCMS's2014EHRIncentiveProgramforEligibleProfessionals:ReleaseNotes10/1/2012CPTonlycopyright2010AmericanMedicalAssociation.
Allrightsreserved.
CPTisaregisteredtrademarkoftheAmericanMedicalAssociation.
ApplicableFARS/DFARSapplytogovernmentuse.
Feeschedules,relativevalueunits,conversionfactors,and/orrelatedcomponentsarenotassignedbytheAMA,arenotpartofCPT,andtheAMAisnotrecommendingtheiruse.
TheAMAdoesnotdirectlyorindirectlypracticemedicineordispensemedicalservices.
TheAMAassumesnoliabilityfordatacontainedornotcontainedherein.
CLINICALQUALITYMEASURESFORCMS'S2014EHRINCENTIVEPROGRAMFORELIGIBLEPROFESSIONALSRELEASENOTESInAugust2012,theCentersforMedicare&MedicaidServices(CMS)finalizedtheclinicalqualitymeasures(CQMs)forthe2014MedicareandMedicaidElectronicHealthRecord(EHR)IncentiveProgramforEligibleProfessionals,alsoknownasMeaningfulUseStage2(MU2)forEligibleProfessionals.
1ThislistofCQMsforMU2includesmeasuresretainedfromMeaningfulUseStage1(MU1)foruseinMU2.
AllretainedMU1measureshavebeenupdatedbasedonadvancesintechnologyandtoolsforeMeasuredevelopment,commentsfromstakeholders,changesinitiatedbymeasuredevelopers,andCMS'sstandardsasdefinedintheagency'sMeasuresManagementSystemBlueprint,Version8(Blueprint).
2CMSrecognizestheimportanceofprovidingsupport,training,andinformationtoMUstakeholders,particularlyastheEHRIncentiveProgramstransitiontoMU2.
ThepurposeofthisdocumentistoinformeligibleprovidersandthevendorcommunityaboutupdatedprogramrequirementsrelatedtotheCQMs.
ThisupdateincludesinformationaboutglobalchangesincorporatedacrossallmeasuresaswellasspecificchangestothemeasuresretainedinMU2.
Globalchangesarelistedfirstandincludestructuralmodifications;updatestovaluesets;anddataelementsandstandardsrevisedinaccordancewiththeBlueprint.
Specificchangestomeasuresincludechangestomeasurecomponents,suchasinitialpatientpopulations,1CMS.
"MedicareandMedicaidPrograms;ElectronicHealthRecordIncentiveProgram—Stage2.
"2012.
Availableat[http://www.
ofr.
gov/(X(1)S(uzclbwrx5fwqm2w2mipkysrh))/OFRUpload/OFRData/2012-21050_PI.
pdf].
AccessedAugust28,2012.
2CMS.
"CMSMeasureManagementSystemBlueprint,Version8.
"Availableat[http://www.
cms.
gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/MMS/MeasuresManagementSystemBlueprint.
html].
AccessedAugust28,2012.
Page2of16denominators,numerators,exclusions,andexceptions,aswellaslogicchangesthataffecthowdataelementsinterrelateduringthemeasurementperiod.
ThisdocumentisintendedforreaderswhoarefamiliarwitheMeasurecomponentsandthecurrentstandardsforconstructinganeMeasure.
FormoreinformationoneMeasures,pleasevisittheCMSwebsite(http://www.
cms.
gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/QualityMeasures/Downloads/QMGuideForReadingEHR.
pdf)anddownloadtheGuideforReadingtheEHRIncentiveProgramEPMeasures.
GlobalEditsIntroducedanewmeasure-identificationschemethatcombinestheeMeasureidentifier,NationalQualityForum(NQF)number(ifapplicable),andeMeasureversionnumber.
Updatedtherationale,clinicalrecommendationstatements,andreferencestoincludethelatestclinicalguidancerelatedtothemeasures.
Providedadditionalguidancetohelpimplementersinterpretthecalculationrequirementsforthemeasures.
UpdatedtheeMeasureheadertoreflectBlueprintrequirements(suchasusingtheinitialpatientpopulationtodefinethedenominatorandincludingstratificationvariablesintheheader)andmodifiedotherfields,suchaspopulationcriteria,toreflectthesechanges.
Changedthestandardizationofthemeasurementperiodfrom"year"to"period.
"UpdatedthemeasurelogictoreflectthechangestotheQualityDataModel(QDM),toreflectconsistentuseofrelativetimingacrossmeasures(includingagecalculation),occurencing,anddenominatorexclusions.
Assigneddataelementsbasedonversion2.
1.
1.
1oftheQDM3toeachclinicalconcept,addingattributesasneededtopreciselydefineQDMelements.
FormeasuresusingtheQDMof"Medication,Active,"addedtheAND/ANDNOTconstructtocompensateforvaryinginterpretationsoftherelativetiming"during.
"The'"Medication,Active"periodcanstartatanytimebutcannotendbefore"OccurrenceAofEncounter,Performed.
"3FormoreontheQualityDataModel,visittheNQFwebsiteathttp://www.
qualityforum.
org/Projects/h/QDS_Model/QDS_Version_2_1.
aspx.
Page3of16Incorporatedsupplementaldataelements(race,ethnicity,sex,andpayer)asrequiredbytheBlueprint.
Reorganizedandretitledtheencountervaluesetstostandardizethemacrossdevelopers.
AlsoincorporatedencountervaluesetsusingSNOMED-CTtoalignwiththeHealthInformationTechnologyStandardsCommittee's(HITSC's)vocabularyrecommendationsfortheQDMdatatype"Encounter.
"Updatedexistingvaluesetsandaddednewvaluesetstoalignwiththetransitionalandfinalvocabularies,basedontheHITSCrecommendationsandrequiredbytheBlueprint.
FullyspecifiedICD-9-CMandICD-10-CMcodesand,asapplicable,ensuredconsistencywiththe2012PhysicianQualityReportingProgram(PQRS)measures.
Providedgroupingobjectidentifiersforeachdataelement.
NQF0002:AppropriateTestingforChildrenwithPharyngitisClarifiedtheinitialpatientpopulationtoonlyincludethereviewthefirstepisodeoftonsillitisorpharyngitisperpatient.
Includedonlyorderedmedicationsasacriteriafortheinitialpatientpopulation.
ModifiedthedenominatorexclusionstoonlyincludeactiveantibioticmedicationsAddedtherestrictionthatantibioticmedicationinthedenominatorexclusionsmustbeactive30daysbeforethediagnosisbecausetheencounterislinkedtothediagnosisthroughtheinitialpatientpopulationcriteria.
Requiredthelaboratorytesttohavearesultpresenttoensurethatthetesthasbeencompleted.
UpdatedthenumeratorcriteriatorequirethelaboratorytestforStreptococcustooccurwithinthreedaysoftheepisode.
NQF0004:InitiationandEngagementofAlcoholandOtherDrugDependenceTreatmentUpdatedthemeasuretitleforconsistencywiththemeasuretitlesusedinotherprograms,suchasthePQRS.
Updatedthemeasuredescriptiontodefine"adolescentandadultpatients"aspatientsage13orolder.
Modifiedthestructureofthemeasuresothatitisasinglemeasurewithreportingstratifiedbyagegroup.
Changedthetimewindowforthefirstdiagnosisofalcoholordrugdependencyintheinitialpatientpopulationtothefirst11monthsofthemeasurementperiod.
Page4of16Updatedthespecificationstorequireuseofdiagnosiscriteriatodetermineifthepatienthasthecondition,andtorequireuseofencountercriteriatoidentifythebeginningoftheepisodebyrequiringthediagnosistostartduringtheencounter.
Removedacuteandnonacuteinpatientencountersfornumerator1.
Clarifiedthat,tobecompliantfornumerator2,thepatientmustmeetthecriteriaforbothnumerator1and2.
NQF0018:ControllingHighBloodPressureClarifiedthatthediagnosisofhypertensioncouldoccuranytimebetweenthefirstpartofthemeasurementperiodtobeforethemeasurementperiod.
Changedtheagerangefromage17–84toage18–85.
Clarifiedthatthedenominatorexclusionofpregnancyhadtobeinthemeasurementperiod.
Clarifiedthatthesystolicanddiastolicbloodpressurereadingmustcomefromthemostrecentvisit.
NQF0024:WeightAssessmentandCounselingforNutritionandPhysicalActivityforChildrenandAdolescentsModifiedthestructureofthemeasuresoitisasinglemeasurewithreportingstratifiedbyagegroups.
Changedtheagecriteriaoftheinitialpatientpopulationfrompatientsage2–17toage3–17.
Clarifiedthattheeligibleencounterintheinitialpatientpopulationshouldbewithaprimarycarephysicianorobstetrician/gynecologist.
Changedthedenominatorexclusiontoonlyincludeadiagnosisofpregnancy.
Addedthepatient'sheightandweighttonumeratorcriteria,inadditiontobodymassindex.
NQF0028:PreventiveCareandScreening:TobaccoUse:ScreeningandCessationInterventionCombinedthemeasurepairaandbintoonemeasure,andupdatedthetitletoreflectthismodification.
Expandedthedefinitionof"eligibleencounters"intheinitialpatientpopulation.
Combinedthenumeratorcriteriafromthepreviouslypairedmeasures.
UpdatedtheQDMdatatypefor"Procedure,Performed:TobaccoUseCessationCounselingto"Intervention,Performed:TobaccoUseCessationCounseling.
"Page5of16Addeddenominatorexceptionsformedicalreasons.
NQF0031:BreastCancerScreeningAdded"female"sexcriteriatotheinitialpatientpopulation.
Changedtheagecriteriaoftheinitialpatientpopulationfromage41–68toage42–69.
Changedtheeligibletimeperiodforanencounterintheinitialpatientpopulationfromtwoyearstoduringtheone-yearmeasurementperiod.
Clarifiedthatbecauseweareonlylookingforcompletemastectomies,apatientthathadtwounilateralmastectomiesshouldbeexcluded.
Changedthenumeratorcriterionfrom"performed"torequiringa"result"tobepresent.
NQF0032:CervicalCancerScreeningAdded"female"sexcriteriatotheinitialpatientpopulation.
Changedtheeligibletimeperiodforanencounterintheinitialpatientpopulationfromthreeyearstoduringtheone-yearmeasurementperiod.
Changedtheagecriteriaoftheinitialpatientpopulationfromage23–63toage24–63.
NQF0033:ChlamydiaScreeningforWomenModifiedthestructureofthemeasuresoitisasinglemeasurewithreportingstratifiedbyagegroup.
Changedtheagecriteriaoftheinitialpatientpopulationfromage15–24toage16–24.
Added"female"sexcriteriatotheinitialpatientpopulation.
Updatedthecategoriesofeventsthatidentifywomenassexuallyactive.
Removedactiveanddispensedmedicationsandperformedproceduresfromthelistofexclusions,requiringonlythattheprocedureormedicationbeordered.
NQF0034:ColorectalCancerScreeningChangedtheeligibletimeperiodforanencounterintheinitialpatientpopulationfromtwoyearstoduringtheone-yearmeasurementperiod.
Changedtheagecriteriaoftheinitialpatientpopulationfromage50–74toage51–75.
Addedanexclusionformalignantneoplasmofthecolon.
Page6of16NQF0036:UseofAppropriateMedicationsforAsthmaChangedtheagecriteriaoftheinitialpatientpopulationfromage5–50toage5–64.
Modifiedthestructureofthemeasuresoitisasinglemeasurewithreportingstratifiedbyagegroups.
Clarifiedtheeligibletimewindowforadiagnosisofpersistentasthmatoanytimebeforeorduringthemeasurementperiod,witharequirementofonlyoneencounter.
Modifiedthecriteriafortheinitialpatientpopulationtorequireadiagnosisofasthma(medicationalonewillnotsuffice).
Removedactiveanddispensedmedicationsfromthelistofnumeratorcriteria,requiringonlythatthemedicationbeordered.
NQF0038:ChildhoodImmunizationStatusModifiedthemeasuretoreportonlyonecombinedrate;separateratesforeachvaccineoracombinationofvaccineswillnolongerbecalculated.
ClarifiedthattheencountercriteriafortheinitialpatientpopulationdoesnotneedtobewithaprimarycareorOB/GYNprovider.
Expandedthenumeratorcriteriatoincludebothmedicationadministeredandtheprocedureforadministeringthevaccine.
Changedtheexclusionfor"medicationallergy"tobedefinedbyananaphylacticreactiontothevaccineandallowedthisreactiontocountasnumeratorcomplianceforeachvaccine.
Combinedtheseparatemeasles,mumps,andrubella(MMR)administeredvaccinesintoonenumeratorcriterion.
AlsoupdatedthetimewindowforMMRvaccineadministrationtooccuranytimebeforethepatient'ssecondbirthdayinthenumerator.
Allowedpastdiagnosesofdiseasetocountfortheappropriatevaccine.
AddedalaboratorytestforthehepatitisAantigentothenumeratorcriteriaforthehepatitisAvaccine.
AlsoupdatedhepatitisAmedicationcriteriatoallowonlyonevaccinationtocountfornumeratorcompliance.
UpdatedtheHiBvaccinemedicationcriteriatorequirethreevaccinationsforcompliance.
Separatedtwo-andthree-doserotavirusvaccinestoensurethepropernumberofdosesisadministered.
Page7of16NQF0041:PreventiveCareandScreening:InfluenzaImmunizationUpdatedthemeasuretitletoreflecttheupdatedmeasurespecifications.
Expandedtheagegrouptoincludeallpatientsage6monthsorolderintheinitialpatientpopulation.
ChangedthetimewindowofthedenominatorforwhichtheencountersmustoccurtoreflectthenewtimeperiodforthefluseasonrecommendedbytheCentersforDiseaseControlandPrevention.
Added"peritonealdialysisprocedure"and"hemodialysisprocedure"tothedenominatorcriteria.
Addedtothenumeratoranycommunicationfrompatienttoproviderregardingthepreviousreceiptofavaccine.
NQF0043:PneumoniaVaccinationStatusforOlderAdultsRestrictedthetimewindowforanencounterintheinitialpatientpopulationtoonlyduringthemeasurementperiod.
Changedtheagecriteriaoftheinitialpatientpopulationfromage64andoldertoage65andolder.
Addedpneumococcalvaccineadministeredandhistoryofapneumococcalvaccinetothenumeratorcriteria.
NQF0052:UseofImagingStudiesforLow-BackPainRephrasedthemeasuretitle.
Changedtheagecriteriaoftheinitialpatientpopulationfromage18–49toage18–50.
Addedtheeligibleageofpatients(18–50)tothemeasuredescription.
Specifiedthatthelow-backpaindiagnosismustoccurduringanofficeoremergency-departmentvisitnomorethan337daysafterthestartofthemeasurementperiod.
Movedtheexclusioncriteriaforthedenominatortothedenominator-exclusionsection,includingalow-backpaindiagnosislessthan180daysbeforeoccurrenceAofalow-backpaindiagnosisoradiagnosisofcancer,trauma,IVdrugabuse,orneurologicimpairmentduringtheyearbeforethemeasurementperiod.
Changedthemeasuretocalculatethenumberofpatientswithadiagnosisoflow-backpainwhodidhaveanimagingstudy(e.
g.
,X-ray,MRI,CTscan)within28daysofthediagnosis;alowerrateisthusabetterscoreforthismeasure.
Page8of16NQF0055:EyeExamChangedtheagecriteriaoftheinitialpatientpopulationfromage18–74toage18–75.
Removedfromthedenominatorcriteriaanydispensed,ordered,oractivemedicationsindicativeofdiabetes.
Updatedthetimewindowforanactivediabetesdiagnosistoanytimebeforeorduringthemeasurementperiod.
Modifiedtheencountercriteriaintheinitialpatientpopulation,includinglimitingthetimewindowtothemeasurementperiod.
Modifiedtheexclusioncriteriaforthedenominator,includingremovingpolycysticovariesdiagnosisandmedicationsindicativeofdiabetesaswellasrestrictingthetimewindowforanactivegestational-diabetesdiagnosistothemeasurementperiod.
Limitedtheeye-examproceduresinthenumeratortoeitheranegativeretinalexamduringtheyearbeforethemeasurementperiodoraretinalordilatedeyeexamduringthemeasurementperiod.
NQF0056:Diabetes:FootExamRemoveddetailsaboutthetypeofdiabetes(type1or2)andtypeoffootexam(visualinspection,sensoryexamwithmonofilament,orpulseexam)fromthemeasuredescription.
Changedtheagecriteriaoftheinitialpatientpopulationfromage18–74toage18–75.
Removedfromthedenominatorcriteriaanydispensed,ordered,oractivemedicationsindicativeofdiabetes.
Updatedthetimewindowforanactivediabetesdiagnosistoanytimebeforeorduringthemeasurementperiod.
Modifiedtheencountercriteriaintheinitialpatientpopulation,includinglimitingthetimewindowtothemeasurementperiod.
Modifiedtheexclusioncriteriaforthedenominator,includingremovingpolycysticovariesandmedicationsindicativeofdiabetesaswellasrestrictingthetimewindowforanactivegestational-diabetesdiagnosistothemeasurementperiod.
Modifiedthefoot-examcriteriatoincludethespecificcomponentsofthefootexam,includingavisualexamandeitherasensoryorpulseexamduringthemeasurementperiod.
Page9of16NQF0059:Diabetes:HemoglobinA1cPoorControlChangedtheagecriteriaoftheinitialpatientpopulationfromage18–74toage18–75.
Removedfromthedenominatorcriteriaanydispensed,ordered,oractivemedicationsindicativeofdiabetes.
Updatedthetimewindowforadiagnosisofactivediabetestoanytimebeforeorduringthemeasurementperiod.
Modifiedtheencountercriteriaintheinitialpatientpopulation,includinglimitingthetimewindowtothemeasurementperiod.
Modifiedtheexclusioncriteriaforthedenominator,includingremovingpolycysticovariesandmedicationsindicativeofdiabetesaswellasrestrictingthetimewindowforanactivegestational-diabetesdiagnosistothemeasurementperiod.
AddedtheabsenceofanHbA1claboratorytestresultsduringthemeasurementperiodforeligiblepatientsasanumeratorcriterion.
NQF0062:Diabetes:UrineProteinScreeningUpdatedthemeasuretitletospecifyproteinscreening.
Removedthedetailsaboutthetypeofdiabetes(type1or2)fromthemeasuredescription.
Changedtheagecriteriaoftheinitialpatientpopulationfromage18–74toage18–75.
Removedfromthedenominatorcriteriaanydispensed,ordered,oractivemedicationsindicativeofdiabetes.
Updatedthetimewindowforadiagnosisofactivediabetestoanytimebeforeorduringthemeasurementperiod.
Modifiedtheencountercriteriaintheinitialpatientpopulation,includinglimitingthetimewindowtothemeasurementperiod.
Modifiedtheexclusioncriteriaforthedenominator,includingremovingpolycysticovariesandmedicationsindicativeofdiabetesaswellasrestrictingthetimewindowforanactivegestational-diabetesdiagnosistothemeasurementperiod.
Page10of16NQF0064:Diabetes:Low-DensityLipoprotein(LDL)ManagementandControlRemoved"andControl"fromthemeasuretitle.
SpecifiedinthemeasuredescriptionthatLDL-Cunder100mg/dLisconsideredadequatelycontrolled.
Changedtheagecriteriaoftheinitialpatientpopulationfromage18–74toage18–75.
Removedfromthedenominatorcriteriaanydispensed,ordered,oractivemedicationsindicativeofdiabetes.
Updatedthetimewindowforanactivediabetesdiagnosistoanytimebeforeorduringthemeasurementperiod.
Modifiedtheencountercriteriaintheinitialpatientpopulation,includinglimitingthetimewindowtothemeasurementperiod.
Modifiedtheexclusioncriteriaforthedenominator,includingremovingpolycysticovariesandmedicationsindicativeofdiabetesaswellasrestrictingthetimewindowforanactivegestational-diabetesdiagnosistothemeasurementperiod.
Removedthescreeningindicator.
NQF0068:IschemicVascularDisease(IVD):UseofAspirinorAnotherAntithromboticUpdatedthemeasuredescriptiontoreflectthechangeinthelook-backperiodandactive-medicationrequirement.
Changedtheagecriteriaoftheinitialpatientpopulationfromage17andoldertoage18andolder.
Changedthe"percutaneoustransluminalcardiacangioplasty"categorytothebroadercategoryof"percutaneouscoronaryinterventions"fortheinitialpatientpopulation.
Changedtheeligibletimeperiodfordiagnosesandproceduresfortheinitialpatientpopulationfrom2to12monthsbeforethemeasurementperiodtotheyearbeforethemeasurementperiod.
Removedtherequirementfromtheinitialpatientpopulationthatthediagnosisandproceduresofinterestneededtooccurduringanencounter.
Limitedthenumeratorcriteriato"medicationactive"(notorderedordispensed),andrequiredthatthemedicationbeactiveatsometimeduringthemeasurementperiod.
Page11of16NQF0070:CoronaryArteryDisease(CAD):Beta-BlockerTherapy—PriorMyocardialInfarction(MI)orLeft-VentricularSystolicDysfunction(LVEF)<40%Revisedthemeasuretitleanddescriptiontoreflectthemostup-to-dateinformationfromthemeasuredeveloper/steward.
Dividedthecalculationofthemeasureintotworatestoreflectthetwodistinctdenominatorpopulations.
Itisexpectedthattheimplementerwillreporteachpopulationscoreseparatelyandatotalscore.
Expandedthedefinitionofeligibleencounterininitialpatientpopulations1and2.
Addedadenominatorpopulation;denominator1includespatientswithaprior(resolved)myocardialinfarction,anddenominator2includespatientswithLVEF<40%.
Clarifiedtherecommendedtypeofbeta-blockertherapyforeachdenominatorpopulationintheguidancestatementandlogic,inaccordancewithupdatedclinicalrecommendations.
Addedtothedenominatorexceptionsadditionalmethodsofcapturingallergiesandintolerances,basedonHITSCrecommendationsNQF0075:IschemicVascularDisease(IVD):CompleteLipidPanelandLDLControlChangedthemeasuretitlefrom"LDL"to"LDL-C.
"SpecifiedinthemeasuredescriptionthatLDL-Cunder100mg/dLisconsideredadequatelycontrolled.
Changedtheagecriteriaoftheinitialpatientpopulationfromage17andoldertoage18andolder.
Changed"percutaneoustransluminalcardiacangioplasty"tothebroadercategoryof"percutaneouscoronaryinterventions"fortheinitialpatientpopulation.
Changedtheeligibletimeperiodfordiagnosesandproceduresfortheinitialpatientpopulationfrom2to12monthsbeforethemeasurementperiodtotheyearbeforethemeasurementperiod.
Removedtherequirementfromtheinitialpatientpopulationthatthediagnosisandproceduresofinterestneededtooccurduringanencounter.
Includedarequirementinthenumerator1criteriathatacompletelipid-paneltestresultispresentoralltheseparatecomponentsofacompletelipidpanelmustbeperformedandhavearesult.
Changednumerator2criteriatoincludeonlyaLDL-Clabtestresult<100mg/dL;removedtheothercomponentsneededtocalculatetheLDL-Cforhightriglycerides.
Page12of16NQF0081:HeartFailure(HF):Angiotensin-ConvertingEnzyme(ACE)InhibitororAngiotensin-ReceptorBlocker(ARB)TherapyforLeft-VentricularSystolicDysfunction(LVSD)Revisedthemeasuredescriptiontoreflectthemostup-to-dateinformationfromthemeasuredeveloper/steward.
RemovedonedenominatoroptionusedtocaptureapatientwithLVSD—"LVFASSMT.
"ChangedalldenominatoroptionsforcapturingapatientwithLVSDfroma"startsbeforestartof…"timingto"startsbeforeorduring….
"Addedtothedenominatorexceptionadditionalmethodsofcapturingallergiesandintolerances,basedonHITSCrecommendations.
Refinedthevaluesetsforthedenominatorexception.
ChangedtheQDMdatatypefor"PatientreasonforACEinhibitororARBdecline"valueset.
NQF0083:HeartFailure(HF):Beta-BlockerTherapyforLeft-VentricularSystolicDysfunction(LVSD)Revisedthemeasuredescriptiontoreflectthemostup-to-dateinformationfromthemeasuredeveloper/steward.
RemovedonedenominatoroptionusedtocaptureapatientwithLVSD—"LVFASSMT.
"ChangedalldenominatoroptionsforcapturingapatientwithLVSDfroma"startsbeforestartof…"timingto"startsbeforeorduring….
"Refinedthevaluesetsforthedenominatorexception.
NQF0086:PrimaryOpen-AngleGlaucoma(POAG):Optic-NerveEvaluationRevisedthemeasuredescriptiontoreflectthemostup-to-dateinformationfromthemeasuredeveloper/steward.
Expandedthe"optic-nerveheadevaluation"tocaptureitstwocomponents:cup-to-discratioandoptic-discexamforstructuralabnormalities.
NQF0088:DiabeticRetinopathy:DocumentationofPresenceorAbsenceofMacularEdemaandLevelofSeverityofRetinopathyRevisedthemeasurelogicofthenumeratorbasedontheupdatedQDMfortypeandcategoryofthenumeratorcriteria.
Page13of16NQF0089:DiabeticRetinopathy:CommunicationwiththePhysicianManagingOngoingDiabetesCareUpdatedthedenominatorcriteriafromperformingamacularorfundusexamtoidentifyingtheresultsofthediagnosticstudy.
Furtherdefinedtheresultsofthemacularexaminthenumeratorbyseparatingthefindingsinto"present"or"absent.
"NQF0105:AntidepressantMedicationManagementShortenedthemeasuretitle.
Updatedthemeasuredescriptiontospecifythetwocalculatedrates.
Changedtheagecriteriaoftheinitialpatientpopulationfrompatientsage18andolder245daysintothemeasurementperiodtoage18andolderatthestartofthemeasurementperiod.
Changedtheeligibletimeperiodforadiagnosisofmajordepressionintheinitialpatientpopulation.
Theoriginaltimeperiodwaslessthan245daysbeforethemeasurementperiodstartstonomorethan245daysbeforethemeasurementperiodends.
Therevisedtimeperiodislessthan180daysbeforethemeasurementperiodstartstonomorethan180daysafterthemeasurementperiodends.
Removedthecriterionthatthedepressiondiagnosismustoccurduringanencounter.
Limitedthecriteriafortheinitialpatientpopulationto"antidepressantmedicationactive"(notorderedordispensed).
Removedadenominatorcriterionthatanotherdiagnosisofdepressionnotoccursoonerthan120daysbeforethediagnosisofdepressionfortheepisodeofinterest.
Addedadenominatorexclusionthatanotherantidepressantmedicationnotbeactivelessthan90daysbeforetheantidepressantmedicationofinterest.
Changedthenumeratorcriteriafornumerator1and2tofocusonthecumulativeamountofmedicationdispensed.
NQF0385:ColonCancer:ChemotherapyforStageIIIColonCancerPatientsRevisedthemeasuretitleanddescriptiontoreflectthemostup-to-dateinformationfromthemeasuredeveloper/steward.
Added"AJCC"tothemeasuretitle.
Revisedthemeasuredescriptiontoincludeanupperlimitof80yearsofage.
Alsorevisedtheinitialpatientpopulationtoincludeallpatientsage18through80withcoloncancer.
Page14of16Removedaninactivediagnosisofcoloncancer(historyofcoloncancer)fromthecriteriafortheinitialpatientpopulation,asthemeasureislimitedtopatientswithafirst-recordeddiagnosisofcoloncancerduringthe12-monthreportingperiod(thatis,duringaneligibleencounterbetweenthepatientandprovider).
Excludedpatientswhoseclinical-stagingprocedurestartedbeforetheactivediagnosisofcoloncancer.
Excludedpatientswhosediagnosisofcoloncancerwasmorethantwoyearsbeforethemeasurementenddate.
Specifiedthatthepatient'sclinical-stagingprocedureresultingin"colondistantmetastasisstatusMO"startedbeforetheeligibleencounter.
Specifiedthetumorsizesandlymph-nodestatusesfollowingtheclinical-stagingprocedurethatareeligibleforinclusioninthedenominator.
Specifiedthetimingoforderingoradministeringchemotherapyeligibleforinclusioninthenumerator.
AlternatemethodsofcapturingallergiesandintolerancesaddedtodenominatorexceptionstoalignwithHITSCrecommendations.
NQF0387:BreastCancer:HormonalTherapyforStageIC-IIICEstrogenReceptor/ProgesteroneReceptor(ER/PR)-PositiveBreastCancerRevisedthemeasuretitlebydeletingtheterm"oncology"toreflectthemostup-to-dateinformationfromthemeasuredeveloper/steward.
Specifiedthatanactivediagnosisofbreastcancertookplacelessthanfiveyearsbeforethepatient-providerencounterfortheinitialpatientpopulation.
Removedaninactivediagnosisofbreastcancer(historyofbreastcancer)fromthecriteriafortheinitialpatientpopulation,asthemeasureislimitedtopatientswithafirst-recordeddiagnosisofbreastcancerwithinthepastfiveyears.
Excludedpatientswhoseclinical-stagingprocedurestartedbeforetheactivediagnosisofbreastcancer.
Specifiedthatthepatient'sclinical-stagingprocedureresultingin"breastdistantmetastasisstatusMO"startedbeforetheeligibleencounter.
RemovedthebreastcancerStageIC-IIICprocedurefromthedenominatorcriteriaandaddedtheclinical-stagingprocedure.
Specifiedthetumorsizesandlymph-nodestatusesfollowingtheclinical-stagingprocedurethatareeligibleforinclusioninthedenominator.
Specifiedtheeligibletimingoftheorderinganddispensingoftamoxifenoraromataseinhibitortherapyfornumeratorinclusion.
Page15of16Revisedthedenominatorexceptionstocaptureclinical-trialparticipantsandremovedadversemedicationeventstoalignwithHITSCrecommendations.
NQF0389:ProstateCancer:AvoidanceofOveruseofBoneScanforStagingLow-RiskProstateCancerPatientsCorrectedprostate-specificcriterionforantigentestresultsfrom<=10mg/dLto<=10ng/mL.
Removed"Procedureresult:AJCCcancerstagelow-riskrecurrenceprostatecancer"fromdenominatorcriteria.
Addedperformanceofa"clinicalstagingprocedure"withresultofprostatecancerprimarytumorsizeT1corT2atodenominatorcriteria.
Added"DiagnosticStudy,Order:BoneScan(Source:'Otherprovider')"tolistofdenominatorexceptions(note:MU1denominatorexclusionsarenowconsidereddenominatorexceptionsinMU2).
NQF0421:PreventiveCareandScreening:BodyMassIndex(BMI)ScreeningandFollow-UpUpdatedthedescriptionandnumeratortoincludetimingguidanceforfollow-upof"BMIoutsideofnormalparameters"toinclude"inthepastsixmonthsorduringthecurrentvisit.
"AddeddefinitionsforBodyMassIndex(BMI),CalculatedBMI,andFollow-UpPlan.
RevisedDenominatorExclusionbydeleting"terminalillness"andsystemreasonsfornotcalculatingBMI,added"patientsreceivingpalliativecare,"andmovedpatientandmedicalreasonfornotcalculatingBMItoDenominatorExceptions.
ClarifiedpatientreasonfornotcalculatingBMItoinclude"ThepatientrefusesBMImeasurement.
"ClarifiedmedicalorotherreasonfornotcalculatingBMItoinclude"IfthereisanyotherreasondocumentedinthemedicalrecordbytheproviderexplainingwhyBMImeasurementwasnotappropriate"OR"Ifthepatientisinanurgentoremergentmedicalsituationwheretimeisoftheessenceandtodelaytreatmentwouldjeopardizethepatient'shealthstatus.
"AddedCareGoalandCommunicationfollow-uptoapplicableaboveandbelownormalBMIfollow-upinterventions.
AddedBMIinterventionsfor"AboveNormalFollow-up,""AboveNormalReferrals,""AboveNormalMedications,""BelowNormalFollow-up,""BelowNormalReferrals,"and"BelowNormalMedications.
"Page16of16
Allrightsreserved.
CPTisaregisteredtrademarkoftheAmericanMedicalAssociation.
ApplicableFARS/DFARSapplytogovernmentuse.
Feeschedules,relativevalueunits,conversionfactors,and/orrelatedcomponentsarenotassignedbytheAMA,arenotpartofCPT,andtheAMAisnotrecommendingtheiruse.
TheAMAdoesnotdirectlyorindirectlypracticemedicineordispensemedicalservices.
TheAMAassumesnoliabilityfordatacontainedornotcontainedherein.
CLINICALQUALITYMEASURESFORCMS'S2014EHRINCENTIVEPROGRAMFORELIGIBLEPROFESSIONALSRELEASENOTESInAugust2012,theCentersforMedicare&MedicaidServices(CMS)finalizedtheclinicalqualitymeasures(CQMs)forthe2014MedicareandMedicaidElectronicHealthRecord(EHR)IncentiveProgramforEligibleProfessionals,alsoknownasMeaningfulUseStage2(MU2)forEligibleProfessionals.
1ThislistofCQMsforMU2includesmeasuresretainedfromMeaningfulUseStage1(MU1)foruseinMU2.
AllretainedMU1measureshavebeenupdatedbasedonadvancesintechnologyandtoolsforeMeasuredevelopment,commentsfromstakeholders,changesinitiatedbymeasuredevelopers,andCMS'sstandardsasdefinedintheagency'sMeasuresManagementSystemBlueprint,Version8(Blueprint).
2CMSrecognizestheimportanceofprovidingsupport,training,andinformationtoMUstakeholders,particularlyastheEHRIncentiveProgramstransitiontoMU2.
ThepurposeofthisdocumentistoinformeligibleprovidersandthevendorcommunityaboutupdatedprogramrequirementsrelatedtotheCQMs.
ThisupdateincludesinformationaboutglobalchangesincorporatedacrossallmeasuresaswellasspecificchangestothemeasuresretainedinMU2.
Globalchangesarelistedfirstandincludestructuralmodifications;updatestovaluesets;anddataelementsandstandardsrevisedinaccordancewiththeBlueprint.
Specificchangestomeasuresincludechangestomeasurecomponents,suchasinitialpatientpopulations,1CMS.
"MedicareandMedicaidPrograms;ElectronicHealthRecordIncentiveProgram—Stage2.
"2012.
Availableat[http://www.
ofr.
gov/(X(1)S(uzclbwrx5fwqm2w2mipkysrh))/OFRUpload/OFRData/2012-21050_PI.
pdf].
AccessedAugust28,2012.
2CMS.
"CMSMeasureManagementSystemBlueprint,Version8.
"Availableat[http://www.
cms.
gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/MMS/MeasuresManagementSystemBlueprint.
html].
AccessedAugust28,2012.
Page2of16denominators,numerators,exclusions,andexceptions,aswellaslogicchangesthataffecthowdataelementsinterrelateduringthemeasurementperiod.
ThisdocumentisintendedforreaderswhoarefamiliarwitheMeasurecomponentsandthecurrentstandardsforconstructinganeMeasure.
FormoreinformationoneMeasures,pleasevisittheCMSwebsite(http://www.
cms.
gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/QualityMeasures/Downloads/QMGuideForReadingEHR.
pdf)anddownloadtheGuideforReadingtheEHRIncentiveProgramEPMeasures.
GlobalEditsIntroducedanewmeasure-identificationschemethatcombinestheeMeasureidentifier,NationalQualityForum(NQF)number(ifapplicable),andeMeasureversionnumber.
Updatedtherationale,clinicalrecommendationstatements,andreferencestoincludethelatestclinicalguidancerelatedtothemeasures.
Providedadditionalguidancetohelpimplementersinterpretthecalculationrequirementsforthemeasures.
UpdatedtheeMeasureheadertoreflectBlueprintrequirements(suchasusingtheinitialpatientpopulationtodefinethedenominatorandincludingstratificationvariablesintheheader)andmodifiedotherfields,suchaspopulationcriteria,toreflectthesechanges.
Changedthestandardizationofthemeasurementperiodfrom"year"to"period.
"UpdatedthemeasurelogictoreflectthechangestotheQualityDataModel(QDM),toreflectconsistentuseofrelativetimingacrossmeasures(includingagecalculation),occurencing,anddenominatorexclusions.
Assigneddataelementsbasedonversion2.
1.
1.
1oftheQDM3toeachclinicalconcept,addingattributesasneededtopreciselydefineQDMelements.
FormeasuresusingtheQDMof"Medication,Active,"addedtheAND/ANDNOTconstructtocompensateforvaryinginterpretationsoftherelativetiming"during.
"The'"Medication,Active"periodcanstartatanytimebutcannotendbefore"OccurrenceAofEncounter,Performed.
"3FormoreontheQualityDataModel,visittheNQFwebsiteathttp://www.
qualityforum.
org/Projects/h/QDS_Model/QDS_Version_2_1.
aspx.
Page3of16Incorporatedsupplementaldataelements(race,ethnicity,sex,andpayer)asrequiredbytheBlueprint.
Reorganizedandretitledtheencountervaluesetstostandardizethemacrossdevelopers.
AlsoincorporatedencountervaluesetsusingSNOMED-CTtoalignwiththeHealthInformationTechnologyStandardsCommittee's(HITSC's)vocabularyrecommendationsfortheQDMdatatype"Encounter.
"Updatedexistingvaluesetsandaddednewvaluesetstoalignwiththetransitionalandfinalvocabularies,basedontheHITSCrecommendationsandrequiredbytheBlueprint.
FullyspecifiedICD-9-CMandICD-10-CMcodesand,asapplicable,ensuredconsistencywiththe2012PhysicianQualityReportingProgram(PQRS)measures.
Providedgroupingobjectidentifiersforeachdataelement.
NQF0002:AppropriateTestingforChildrenwithPharyngitisClarifiedtheinitialpatientpopulationtoonlyincludethereviewthefirstepisodeoftonsillitisorpharyngitisperpatient.
Includedonlyorderedmedicationsasacriteriafortheinitialpatientpopulation.
ModifiedthedenominatorexclusionstoonlyincludeactiveantibioticmedicationsAddedtherestrictionthatantibioticmedicationinthedenominatorexclusionsmustbeactive30daysbeforethediagnosisbecausetheencounterislinkedtothediagnosisthroughtheinitialpatientpopulationcriteria.
Requiredthelaboratorytesttohavearesultpresenttoensurethatthetesthasbeencompleted.
UpdatedthenumeratorcriteriatorequirethelaboratorytestforStreptococcustooccurwithinthreedaysoftheepisode.
NQF0004:InitiationandEngagementofAlcoholandOtherDrugDependenceTreatmentUpdatedthemeasuretitleforconsistencywiththemeasuretitlesusedinotherprograms,suchasthePQRS.
Updatedthemeasuredescriptiontodefine"adolescentandadultpatients"aspatientsage13orolder.
Modifiedthestructureofthemeasuresothatitisasinglemeasurewithreportingstratifiedbyagegroup.
Changedthetimewindowforthefirstdiagnosisofalcoholordrugdependencyintheinitialpatientpopulationtothefirst11monthsofthemeasurementperiod.
Page4of16Updatedthespecificationstorequireuseofdiagnosiscriteriatodetermineifthepatienthasthecondition,andtorequireuseofencountercriteriatoidentifythebeginningoftheepisodebyrequiringthediagnosistostartduringtheencounter.
Removedacuteandnonacuteinpatientencountersfornumerator1.
Clarifiedthat,tobecompliantfornumerator2,thepatientmustmeetthecriteriaforbothnumerator1and2.
NQF0018:ControllingHighBloodPressureClarifiedthatthediagnosisofhypertensioncouldoccuranytimebetweenthefirstpartofthemeasurementperiodtobeforethemeasurementperiod.
Changedtheagerangefromage17–84toage18–85.
Clarifiedthatthedenominatorexclusionofpregnancyhadtobeinthemeasurementperiod.
Clarifiedthatthesystolicanddiastolicbloodpressurereadingmustcomefromthemostrecentvisit.
NQF0024:WeightAssessmentandCounselingforNutritionandPhysicalActivityforChildrenandAdolescentsModifiedthestructureofthemeasuresoitisasinglemeasurewithreportingstratifiedbyagegroups.
Changedtheagecriteriaoftheinitialpatientpopulationfrompatientsage2–17toage3–17.
Clarifiedthattheeligibleencounterintheinitialpatientpopulationshouldbewithaprimarycarephysicianorobstetrician/gynecologist.
Changedthedenominatorexclusiontoonlyincludeadiagnosisofpregnancy.
Addedthepatient'sheightandweighttonumeratorcriteria,inadditiontobodymassindex.
NQF0028:PreventiveCareandScreening:TobaccoUse:ScreeningandCessationInterventionCombinedthemeasurepairaandbintoonemeasure,andupdatedthetitletoreflectthismodification.
Expandedthedefinitionof"eligibleencounters"intheinitialpatientpopulation.
Combinedthenumeratorcriteriafromthepreviouslypairedmeasures.
UpdatedtheQDMdatatypefor"Procedure,Performed:TobaccoUseCessationCounselingto"Intervention,Performed:TobaccoUseCessationCounseling.
"Page5of16Addeddenominatorexceptionsformedicalreasons.
NQF0031:BreastCancerScreeningAdded"female"sexcriteriatotheinitialpatientpopulation.
Changedtheagecriteriaoftheinitialpatientpopulationfromage41–68toage42–69.
Changedtheeligibletimeperiodforanencounterintheinitialpatientpopulationfromtwoyearstoduringtheone-yearmeasurementperiod.
Clarifiedthatbecauseweareonlylookingforcompletemastectomies,apatientthathadtwounilateralmastectomiesshouldbeexcluded.
Changedthenumeratorcriterionfrom"performed"torequiringa"result"tobepresent.
NQF0032:CervicalCancerScreeningAdded"female"sexcriteriatotheinitialpatientpopulation.
Changedtheeligibletimeperiodforanencounterintheinitialpatientpopulationfromthreeyearstoduringtheone-yearmeasurementperiod.
Changedtheagecriteriaoftheinitialpatientpopulationfromage23–63toage24–63.
NQF0033:ChlamydiaScreeningforWomenModifiedthestructureofthemeasuresoitisasinglemeasurewithreportingstratifiedbyagegroup.
Changedtheagecriteriaoftheinitialpatientpopulationfromage15–24toage16–24.
Added"female"sexcriteriatotheinitialpatientpopulation.
Updatedthecategoriesofeventsthatidentifywomenassexuallyactive.
Removedactiveanddispensedmedicationsandperformedproceduresfromthelistofexclusions,requiringonlythattheprocedureormedicationbeordered.
NQF0034:ColorectalCancerScreeningChangedtheeligibletimeperiodforanencounterintheinitialpatientpopulationfromtwoyearstoduringtheone-yearmeasurementperiod.
Changedtheagecriteriaoftheinitialpatientpopulationfromage50–74toage51–75.
Addedanexclusionformalignantneoplasmofthecolon.
Page6of16NQF0036:UseofAppropriateMedicationsforAsthmaChangedtheagecriteriaoftheinitialpatientpopulationfromage5–50toage5–64.
Modifiedthestructureofthemeasuresoitisasinglemeasurewithreportingstratifiedbyagegroups.
Clarifiedtheeligibletimewindowforadiagnosisofpersistentasthmatoanytimebeforeorduringthemeasurementperiod,witharequirementofonlyoneencounter.
Modifiedthecriteriafortheinitialpatientpopulationtorequireadiagnosisofasthma(medicationalonewillnotsuffice).
Removedactiveanddispensedmedicationsfromthelistofnumeratorcriteria,requiringonlythatthemedicationbeordered.
NQF0038:ChildhoodImmunizationStatusModifiedthemeasuretoreportonlyonecombinedrate;separateratesforeachvaccineoracombinationofvaccineswillnolongerbecalculated.
ClarifiedthattheencountercriteriafortheinitialpatientpopulationdoesnotneedtobewithaprimarycareorOB/GYNprovider.
Expandedthenumeratorcriteriatoincludebothmedicationadministeredandtheprocedureforadministeringthevaccine.
Changedtheexclusionfor"medicationallergy"tobedefinedbyananaphylacticreactiontothevaccineandallowedthisreactiontocountasnumeratorcomplianceforeachvaccine.
Combinedtheseparatemeasles,mumps,andrubella(MMR)administeredvaccinesintoonenumeratorcriterion.
AlsoupdatedthetimewindowforMMRvaccineadministrationtooccuranytimebeforethepatient'ssecondbirthdayinthenumerator.
Allowedpastdiagnosesofdiseasetocountfortheappropriatevaccine.
AddedalaboratorytestforthehepatitisAantigentothenumeratorcriteriaforthehepatitisAvaccine.
AlsoupdatedhepatitisAmedicationcriteriatoallowonlyonevaccinationtocountfornumeratorcompliance.
UpdatedtheHiBvaccinemedicationcriteriatorequirethreevaccinationsforcompliance.
Separatedtwo-andthree-doserotavirusvaccinestoensurethepropernumberofdosesisadministered.
Page7of16NQF0041:PreventiveCareandScreening:InfluenzaImmunizationUpdatedthemeasuretitletoreflecttheupdatedmeasurespecifications.
Expandedtheagegrouptoincludeallpatientsage6monthsorolderintheinitialpatientpopulation.
ChangedthetimewindowofthedenominatorforwhichtheencountersmustoccurtoreflectthenewtimeperiodforthefluseasonrecommendedbytheCentersforDiseaseControlandPrevention.
Added"peritonealdialysisprocedure"and"hemodialysisprocedure"tothedenominatorcriteria.
Addedtothenumeratoranycommunicationfrompatienttoproviderregardingthepreviousreceiptofavaccine.
NQF0043:PneumoniaVaccinationStatusforOlderAdultsRestrictedthetimewindowforanencounterintheinitialpatientpopulationtoonlyduringthemeasurementperiod.
Changedtheagecriteriaoftheinitialpatientpopulationfromage64andoldertoage65andolder.
Addedpneumococcalvaccineadministeredandhistoryofapneumococcalvaccinetothenumeratorcriteria.
NQF0052:UseofImagingStudiesforLow-BackPainRephrasedthemeasuretitle.
Changedtheagecriteriaoftheinitialpatientpopulationfromage18–49toage18–50.
Addedtheeligibleageofpatients(18–50)tothemeasuredescription.
Specifiedthatthelow-backpaindiagnosismustoccurduringanofficeoremergency-departmentvisitnomorethan337daysafterthestartofthemeasurementperiod.
Movedtheexclusioncriteriaforthedenominatortothedenominator-exclusionsection,includingalow-backpaindiagnosislessthan180daysbeforeoccurrenceAofalow-backpaindiagnosisoradiagnosisofcancer,trauma,IVdrugabuse,orneurologicimpairmentduringtheyearbeforethemeasurementperiod.
Changedthemeasuretocalculatethenumberofpatientswithadiagnosisoflow-backpainwhodidhaveanimagingstudy(e.
g.
,X-ray,MRI,CTscan)within28daysofthediagnosis;alowerrateisthusabetterscoreforthismeasure.
Page8of16NQF0055:EyeExamChangedtheagecriteriaoftheinitialpatientpopulationfromage18–74toage18–75.
Removedfromthedenominatorcriteriaanydispensed,ordered,oractivemedicationsindicativeofdiabetes.
Updatedthetimewindowforanactivediabetesdiagnosistoanytimebeforeorduringthemeasurementperiod.
Modifiedtheencountercriteriaintheinitialpatientpopulation,includinglimitingthetimewindowtothemeasurementperiod.
Modifiedtheexclusioncriteriaforthedenominator,includingremovingpolycysticovariesdiagnosisandmedicationsindicativeofdiabetesaswellasrestrictingthetimewindowforanactivegestational-diabetesdiagnosistothemeasurementperiod.
Limitedtheeye-examproceduresinthenumeratortoeitheranegativeretinalexamduringtheyearbeforethemeasurementperiodoraretinalordilatedeyeexamduringthemeasurementperiod.
NQF0056:Diabetes:FootExamRemoveddetailsaboutthetypeofdiabetes(type1or2)andtypeoffootexam(visualinspection,sensoryexamwithmonofilament,orpulseexam)fromthemeasuredescription.
Changedtheagecriteriaoftheinitialpatientpopulationfromage18–74toage18–75.
Removedfromthedenominatorcriteriaanydispensed,ordered,oractivemedicationsindicativeofdiabetes.
Updatedthetimewindowforanactivediabetesdiagnosistoanytimebeforeorduringthemeasurementperiod.
Modifiedtheencountercriteriaintheinitialpatientpopulation,includinglimitingthetimewindowtothemeasurementperiod.
Modifiedtheexclusioncriteriaforthedenominator,includingremovingpolycysticovariesandmedicationsindicativeofdiabetesaswellasrestrictingthetimewindowforanactivegestational-diabetesdiagnosistothemeasurementperiod.
Modifiedthefoot-examcriteriatoincludethespecificcomponentsofthefootexam,includingavisualexamandeitherasensoryorpulseexamduringthemeasurementperiod.
Page9of16NQF0059:Diabetes:HemoglobinA1cPoorControlChangedtheagecriteriaoftheinitialpatientpopulationfromage18–74toage18–75.
Removedfromthedenominatorcriteriaanydispensed,ordered,oractivemedicationsindicativeofdiabetes.
Updatedthetimewindowforadiagnosisofactivediabetestoanytimebeforeorduringthemeasurementperiod.
Modifiedtheencountercriteriaintheinitialpatientpopulation,includinglimitingthetimewindowtothemeasurementperiod.
Modifiedtheexclusioncriteriaforthedenominator,includingremovingpolycysticovariesandmedicationsindicativeofdiabetesaswellasrestrictingthetimewindowforanactivegestational-diabetesdiagnosistothemeasurementperiod.
AddedtheabsenceofanHbA1claboratorytestresultsduringthemeasurementperiodforeligiblepatientsasanumeratorcriterion.
NQF0062:Diabetes:UrineProteinScreeningUpdatedthemeasuretitletospecifyproteinscreening.
Removedthedetailsaboutthetypeofdiabetes(type1or2)fromthemeasuredescription.
Changedtheagecriteriaoftheinitialpatientpopulationfromage18–74toage18–75.
Removedfromthedenominatorcriteriaanydispensed,ordered,oractivemedicationsindicativeofdiabetes.
Updatedthetimewindowforadiagnosisofactivediabetestoanytimebeforeorduringthemeasurementperiod.
Modifiedtheencountercriteriaintheinitialpatientpopulation,includinglimitingthetimewindowtothemeasurementperiod.
Modifiedtheexclusioncriteriaforthedenominator,includingremovingpolycysticovariesandmedicationsindicativeofdiabetesaswellasrestrictingthetimewindowforanactivegestational-diabetesdiagnosistothemeasurementperiod.
Page10of16NQF0064:Diabetes:Low-DensityLipoprotein(LDL)ManagementandControlRemoved"andControl"fromthemeasuretitle.
SpecifiedinthemeasuredescriptionthatLDL-Cunder100mg/dLisconsideredadequatelycontrolled.
Changedtheagecriteriaoftheinitialpatientpopulationfromage18–74toage18–75.
Removedfromthedenominatorcriteriaanydispensed,ordered,oractivemedicationsindicativeofdiabetes.
Updatedthetimewindowforanactivediabetesdiagnosistoanytimebeforeorduringthemeasurementperiod.
Modifiedtheencountercriteriaintheinitialpatientpopulation,includinglimitingthetimewindowtothemeasurementperiod.
Modifiedtheexclusioncriteriaforthedenominator,includingremovingpolycysticovariesandmedicationsindicativeofdiabetesaswellasrestrictingthetimewindowforanactivegestational-diabetesdiagnosistothemeasurementperiod.
Removedthescreeningindicator.
NQF0068:IschemicVascularDisease(IVD):UseofAspirinorAnotherAntithromboticUpdatedthemeasuredescriptiontoreflectthechangeinthelook-backperiodandactive-medicationrequirement.
Changedtheagecriteriaoftheinitialpatientpopulationfromage17andoldertoage18andolder.
Changedthe"percutaneoustransluminalcardiacangioplasty"categorytothebroadercategoryof"percutaneouscoronaryinterventions"fortheinitialpatientpopulation.
Changedtheeligibletimeperiodfordiagnosesandproceduresfortheinitialpatientpopulationfrom2to12monthsbeforethemeasurementperiodtotheyearbeforethemeasurementperiod.
Removedtherequirementfromtheinitialpatientpopulationthatthediagnosisandproceduresofinterestneededtooccurduringanencounter.
Limitedthenumeratorcriteriato"medicationactive"(notorderedordispensed),andrequiredthatthemedicationbeactiveatsometimeduringthemeasurementperiod.
Page11of16NQF0070:CoronaryArteryDisease(CAD):Beta-BlockerTherapy—PriorMyocardialInfarction(MI)orLeft-VentricularSystolicDysfunction(LVEF)<40%Revisedthemeasuretitleanddescriptiontoreflectthemostup-to-dateinformationfromthemeasuredeveloper/steward.
Dividedthecalculationofthemeasureintotworatestoreflectthetwodistinctdenominatorpopulations.
Itisexpectedthattheimplementerwillreporteachpopulationscoreseparatelyandatotalscore.
Expandedthedefinitionofeligibleencounterininitialpatientpopulations1and2.
Addedadenominatorpopulation;denominator1includespatientswithaprior(resolved)myocardialinfarction,anddenominator2includespatientswithLVEF<40%.
Clarifiedtherecommendedtypeofbeta-blockertherapyforeachdenominatorpopulationintheguidancestatementandlogic,inaccordancewithupdatedclinicalrecommendations.
Addedtothedenominatorexceptionsadditionalmethodsofcapturingallergiesandintolerances,basedonHITSCrecommendationsNQF0075:IschemicVascularDisease(IVD):CompleteLipidPanelandLDLControlChangedthemeasuretitlefrom"LDL"to"LDL-C.
"SpecifiedinthemeasuredescriptionthatLDL-Cunder100mg/dLisconsideredadequatelycontrolled.
Changedtheagecriteriaoftheinitialpatientpopulationfromage17andoldertoage18andolder.
Changed"percutaneoustransluminalcardiacangioplasty"tothebroadercategoryof"percutaneouscoronaryinterventions"fortheinitialpatientpopulation.
Changedtheeligibletimeperiodfordiagnosesandproceduresfortheinitialpatientpopulationfrom2to12monthsbeforethemeasurementperiodtotheyearbeforethemeasurementperiod.
Removedtherequirementfromtheinitialpatientpopulationthatthediagnosisandproceduresofinterestneededtooccurduringanencounter.
Includedarequirementinthenumerator1criteriathatacompletelipid-paneltestresultispresentoralltheseparatecomponentsofacompletelipidpanelmustbeperformedandhavearesult.
Changednumerator2criteriatoincludeonlyaLDL-Clabtestresult<100mg/dL;removedtheothercomponentsneededtocalculatetheLDL-Cforhightriglycerides.
Page12of16NQF0081:HeartFailure(HF):Angiotensin-ConvertingEnzyme(ACE)InhibitororAngiotensin-ReceptorBlocker(ARB)TherapyforLeft-VentricularSystolicDysfunction(LVSD)Revisedthemeasuredescriptiontoreflectthemostup-to-dateinformationfromthemeasuredeveloper/steward.
RemovedonedenominatoroptionusedtocaptureapatientwithLVSD—"LVFASSMT.
"ChangedalldenominatoroptionsforcapturingapatientwithLVSDfroma"startsbeforestartof…"timingto"startsbeforeorduring….
"Addedtothedenominatorexceptionadditionalmethodsofcapturingallergiesandintolerances,basedonHITSCrecommendations.
Refinedthevaluesetsforthedenominatorexception.
ChangedtheQDMdatatypefor"PatientreasonforACEinhibitororARBdecline"valueset.
NQF0083:HeartFailure(HF):Beta-BlockerTherapyforLeft-VentricularSystolicDysfunction(LVSD)Revisedthemeasuredescriptiontoreflectthemostup-to-dateinformationfromthemeasuredeveloper/steward.
RemovedonedenominatoroptionusedtocaptureapatientwithLVSD—"LVFASSMT.
"ChangedalldenominatoroptionsforcapturingapatientwithLVSDfroma"startsbeforestartof…"timingto"startsbeforeorduring….
"Refinedthevaluesetsforthedenominatorexception.
NQF0086:PrimaryOpen-AngleGlaucoma(POAG):Optic-NerveEvaluationRevisedthemeasuredescriptiontoreflectthemostup-to-dateinformationfromthemeasuredeveloper/steward.
Expandedthe"optic-nerveheadevaluation"tocaptureitstwocomponents:cup-to-discratioandoptic-discexamforstructuralabnormalities.
NQF0088:DiabeticRetinopathy:DocumentationofPresenceorAbsenceofMacularEdemaandLevelofSeverityofRetinopathyRevisedthemeasurelogicofthenumeratorbasedontheupdatedQDMfortypeandcategoryofthenumeratorcriteria.
Page13of16NQF0089:DiabeticRetinopathy:CommunicationwiththePhysicianManagingOngoingDiabetesCareUpdatedthedenominatorcriteriafromperformingamacularorfundusexamtoidentifyingtheresultsofthediagnosticstudy.
Furtherdefinedtheresultsofthemacularexaminthenumeratorbyseparatingthefindingsinto"present"or"absent.
"NQF0105:AntidepressantMedicationManagementShortenedthemeasuretitle.
Updatedthemeasuredescriptiontospecifythetwocalculatedrates.
Changedtheagecriteriaoftheinitialpatientpopulationfrompatientsage18andolder245daysintothemeasurementperiodtoage18andolderatthestartofthemeasurementperiod.
Changedtheeligibletimeperiodforadiagnosisofmajordepressionintheinitialpatientpopulation.
Theoriginaltimeperiodwaslessthan245daysbeforethemeasurementperiodstartstonomorethan245daysbeforethemeasurementperiodends.
Therevisedtimeperiodislessthan180daysbeforethemeasurementperiodstartstonomorethan180daysafterthemeasurementperiodends.
Removedthecriterionthatthedepressiondiagnosismustoccurduringanencounter.
Limitedthecriteriafortheinitialpatientpopulationto"antidepressantmedicationactive"(notorderedordispensed).
Removedadenominatorcriterionthatanotherdiagnosisofdepressionnotoccursoonerthan120daysbeforethediagnosisofdepressionfortheepisodeofinterest.
Addedadenominatorexclusionthatanotherantidepressantmedicationnotbeactivelessthan90daysbeforetheantidepressantmedicationofinterest.
Changedthenumeratorcriteriafornumerator1and2tofocusonthecumulativeamountofmedicationdispensed.
NQF0385:ColonCancer:ChemotherapyforStageIIIColonCancerPatientsRevisedthemeasuretitleanddescriptiontoreflectthemostup-to-dateinformationfromthemeasuredeveloper/steward.
Added"AJCC"tothemeasuretitle.
Revisedthemeasuredescriptiontoincludeanupperlimitof80yearsofage.
Alsorevisedtheinitialpatientpopulationtoincludeallpatientsage18through80withcoloncancer.
Page14of16Removedaninactivediagnosisofcoloncancer(historyofcoloncancer)fromthecriteriafortheinitialpatientpopulation,asthemeasureislimitedtopatientswithafirst-recordeddiagnosisofcoloncancerduringthe12-monthreportingperiod(thatis,duringaneligibleencounterbetweenthepatientandprovider).
Excludedpatientswhoseclinical-stagingprocedurestartedbeforetheactivediagnosisofcoloncancer.
Excludedpatientswhosediagnosisofcoloncancerwasmorethantwoyearsbeforethemeasurementenddate.
Specifiedthatthepatient'sclinical-stagingprocedureresultingin"colondistantmetastasisstatusMO"startedbeforetheeligibleencounter.
Specifiedthetumorsizesandlymph-nodestatusesfollowingtheclinical-stagingprocedurethatareeligibleforinclusioninthedenominator.
Specifiedthetimingoforderingoradministeringchemotherapyeligibleforinclusioninthenumerator.
AlternatemethodsofcapturingallergiesandintolerancesaddedtodenominatorexceptionstoalignwithHITSCrecommendations.
NQF0387:BreastCancer:HormonalTherapyforStageIC-IIICEstrogenReceptor/ProgesteroneReceptor(ER/PR)-PositiveBreastCancerRevisedthemeasuretitlebydeletingtheterm"oncology"toreflectthemostup-to-dateinformationfromthemeasuredeveloper/steward.
Specifiedthatanactivediagnosisofbreastcancertookplacelessthanfiveyearsbeforethepatient-providerencounterfortheinitialpatientpopulation.
Removedaninactivediagnosisofbreastcancer(historyofbreastcancer)fromthecriteriafortheinitialpatientpopulation,asthemeasureislimitedtopatientswithafirst-recordeddiagnosisofbreastcancerwithinthepastfiveyears.
Excludedpatientswhoseclinical-stagingprocedurestartedbeforetheactivediagnosisofbreastcancer.
Specifiedthatthepatient'sclinical-stagingprocedureresultingin"breastdistantmetastasisstatusMO"startedbeforetheeligibleencounter.
RemovedthebreastcancerStageIC-IIICprocedurefromthedenominatorcriteriaandaddedtheclinical-stagingprocedure.
Specifiedthetumorsizesandlymph-nodestatusesfollowingtheclinical-stagingprocedurethatareeligibleforinclusioninthedenominator.
Specifiedtheeligibletimingoftheorderinganddispensingoftamoxifenoraromataseinhibitortherapyfornumeratorinclusion.
Page15of16Revisedthedenominatorexceptionstocaptureclinical-trialparticipantsandremovedadversemedicationeventstoalignwithHITSCrecommendations.
NQF0389:ProstateCancer:AvoidanceofOveruseofBoneScanforStagingLow-RiskProstateCancerPatientsCorrectedprostate-specificcriterionforantigentestresultsfrom<=10mg/dLto<=10ng/mL.
Removed"Procedureresult:AJCCcancerstagelow-riskrecurrenceprostatecancer"fromdenominatorcriteria.
Addedperformanceofa"clinicalstagingprocedure"withresultofprostatecancerprimarytumorsizeT1corT2atodenominatorcriteria.
Added"DiagnosticStudy,Order:BoneScan(Source:'Otherprovider')"tolistofdenominatorexceptions(note:MU1denominatorexclusionsarenowconsidereddenominatorexceptionsinMU2).
NQF0421:PreventiveCareandScreening:BodyMassIndex(BMI)ScreeningandFollow-UpUpdatedthedescriptionandnumeratortoincludetimingguidanceforfollow-upof"BMIoutsideofnormalparameters"toinclude"inthepastsixmonthsorduringthecurrentvisit.
"AddeddefinitionsforBodyMassIndex(BMI),CalculatedBMI,andFollow-UpPlan.
RevisedDenominatorExclusionbydeleting"terminalillness"andsystemreasonsfornotcalculatingBMI,added"patientsreceivingpalliativecare,"andmovedpatientandmedicalreasonfornotcalculatingBMItoDenominatorExceptions.
ClarifiedpatientreasonfornotcalculatingBMItoinclude"ThepatientrefusesBMImeasurement.
"ClarifiedmedicalorotherreasonfornotcalculatingBMItoinclude"IfthereisanyotherreasondocumentedinthemedicalrecordbytheproviderexplainingwhyBMImeasurementwasnotappropriate"OR"Ifthepatientisinanurgentoremergentmedicalsituationwheretimeisoftheessenceandtodelaytreatmentwouldjeopardizethepatient'shealthstatus.
"AddedCareGoalandCommunicationfollow-uptoapplicableaboveandbelownormalBMIfollow-upinterventions.
AddedBMIinterventionsfor"AboveNormalFollow-up,""AboveNormalReferrals,""AboveNormalMedications,""BelowNormalFollow-up,""BelowNormalReferrals,"and"BelowNormalMedications.
"Page16of16
- recommendations科讯cms相关文档
- Surgery科讯cms
- hospitalization科讯cms
- Carriers科讯cms
- Follow科讯cms
- Privigen科讯cms
- 1.0科讯cms
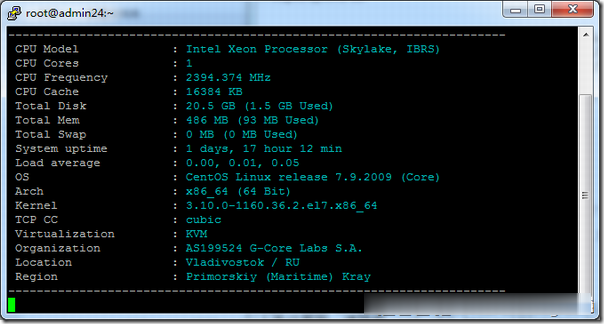
Gcore(gcorelabs)俄罗斯海参崴VPS简单测试
有一段时间没有分享Gcore(gcorelabs)的信息了,这是一家成立于2011年的国外主机商,总部位于卢森堡,主要提供VPS主机和独立服务器租用等,数据中心包括俄罗斯、美国、日本、韩国、新加坡、荷兰、中国(香港)等多个国家和地区的十几个机房,商家针对不同系列的产品分为不同管理系统,比如VPS(Hosting)、Cloud等都是独立的用户中心体系,部落分享的主要是商家的Hosting(Virtu...
HTTPS加密协议端口默认是多少且是否支持更换端口访问
看到群里网友们在讨论由于不清楚的原因,有同学的网站无法访问。他的网站是没有用HTTPS的,直接访问他的HTTP是无法访问的,通过PING测试可以看到解析地址已经比较乱,应该是所谓的DNS污染。其中有网友提到采用HTTPS加密证书试试。因为HTTP和HTTPS走的不是一个端口,之前有网友这样测试过是可以缓解这样的问题。这样通过将网站绑定设置HTTPS之后,是可以打开的,看来网站的80端口出现问题,而...

美国200G美国高防服务器16G,800元
美国高防服务器提速啦专业提供美国高防服务器,美国高防服务器租用,美国抗攻击服务器,高防御美国服务器租用等。我们的海外高防服务器带给您坚不可摧的DDoS防护,保障您的业务不受攻击影响。HostEase美国高防服务器位于加州和洛杉矶数据中心,均为国内访问速度最快最稳定的美国抗攻击机房,带给您快速的访问体验。我们的高防服务器配有最高层级的DDoS防护系统,每款抗攻击服务器均拥有免费DDoS防护额度,让您...

科讯cms为你推荐
-
搜狗360360浏览器为什么不能让我自动登录了360退出北京时间utc+8 13:30-14:00换成北京时间是什么时候重庆电信断网电信光纤一直掉线,打电话问说是机房出了问题 要排查,已经一个星期了还没弄好,大概需要多久才能弄好?中国企业在线用什么软件查找中国所有企业名称360邮箱免费注册360账号-电子邮箱怎么填写?flashftp下载《蔓蔓青萝(全)》.TXT_微盘下载申请支付宝账户怎样申请支付宝账户?要填写什么信息?爱买网超爱买网的特点400电话查询如何辨别400电话的真伪?dezender如何将shopex和phpwind两个伪静态规则写在一起