cm003hh.com
003hh.com 时间:2021-04-07 阅读:()
Determinationofmercurylevelsinenvironmentalsamplesusing2-acetylfuranisonicotinoylhydrazonebyspectrophotometricmethodFullPaperINTRODUCTIONTheanalyticalmonitoringofmercuryinenvironmen-tal,biological,industrialandfoodsamplesisextremelyimportantbecauseofthehightoxicityofthismetalbothinitsinorganicandorganiccompounds[1].
Anexampleforacutemercurypoisoningis"Mina-matadiseases"whichcausesmentaldisturbance,alossofbalance,speech,sightandhearing,difficultyinswallowingandfinallycomaanddeath[2].
Thetoxicityofmercuryde-pendsonitschemicalnature.
Inorganicmercuryhasaveryhighaffinityforproteinsulf-hydrylgroups,accu-mulatesinkidneys,whereasorganicmercuryismoreV.
SaleemBasha1,S.
VidyasagarBabu2,G.
Narasimha3,K.
HussainReddy2*1GovernmentDegreeCollege(Men),Anantapur,A.
P,(INDIA)2DepartmentofChemistry,SriKrishnadevarayaUniversity,Anantapur–515003.
A.
P,(INDIA)3AppliedMicrobiologylaboratory,DepartmentofVirology,SriVenkateswaraUniversity,Tirupati-517502AndhraPradesh,(INDIA)E-mail:khussainreddy@yahoo.
co.
in.
comtoxicsinceitissolubleinfat,thelipidfractionsofmem-branes,andbraintissue.
Mercurytoxicityiscausedmainlybythefactthatitentersthelivingorganismandreactswithdifferentenzymesinhibitingthecatalysisofbasicmetabolicreactions[3].
Theabilityoflivingorgan-ismstoconvertinorganicmercurytoorganicmercurycompoundswhicharemoretoxicandaccumulatetoagreaterextentinsensitivetissues,isalsoaconsiderablefact.
Themainsourcesofmercuryfromwhichitaccu-mulatesintheenvironmentaresyntheticadhesives,airconditionerfilters,amalgams,autoexhausts,anatomicpreservatives,antifoulingpaints,dentalamalgams,bat-teries,cathodetubes,bloodbanksaline,cinnabaretc.
KEYWORDSBiological;Soil;Environmentalsamples;Mercury(II);Substitutedhydrazones;Spectrophotometry.
ABSTRACT2-acetylfuranisonicotinoylhydrazone(AFINH)hasbeensynthesizedandproposedasanewchromogenicreagentforarapid,simple,selective,directandnon-extractivespectrophotometricmethodisdevelopedforthedeterminationofmercury(II)inaqueousdimethylformamide(DMF).
Thereagentgivesdeepyellowcoloured,1:1(M:L)complexwithmercury(II)insodiumacetate-aceticacidbuffermediumofpH5.
5atmax365nm.
Thecolourreactionisinstantaneousandtheabsorbanceremainsconstantforabout12hrs.
Themolarabsorptivityofmercurycomplexis2.
3x103Lmol-1cm-1andsandell'ssensitivityarefoundtobe0.
0888gcm-2.
Themethodwassuccessfullyappliedtoanumberofenvironmental,biologicalandsoilsamples.
Theresultswerecomparablewiththoseobtainedbydithizonemethod.
Theproposedsystemproducedsatisfactoryresultsforthedeter-minationofHg(II).
Theresultsarequiteencouragingandcanbringaware-nessamongthepublic.
2013TradeScienceInc.
-INDIAACAIJ,13(3)2013[81-85]AnIndianJournalVolume13Issue3AnalyticalCHEMISTRYAnalyticalCHEMISTRYISSN:0974-7419.
82Determinationofmercurylevelsinenvironmentalsamplesusing2-acetylfuranFullPaperACAIJ,13(3)2013K.
HussainReddyetal.
83FullPaperACAIJ,13(3)2013AnIndianJournalAnalyticalCHEMISTRYAnalyticalCHEMISTRYDeterminationofmercury(II)Mercury(II)reactswithAFINHinacidicpH(5.
5)togivecolouredcomplexes.
Thecolourreactionisin-stantaneousevenatroomtemperature.
Theorderofadditionofreagent,metalion,buffer,2.
5mlofDMF.
Theabsorbanceofthecolouredcomplexremainscon-stantformorethan2hours.
A10-foldmolarexcessofthereagentisadequateforfullcolourdevelopment.
Ad-ditionofexcessofreagenthasnoadverseeffectontheabsorbanceofthecomplexes.
ThesystemobeysBeer'slawintheconcentrationrange3.
21-32.
1g/mlofmer-cury.
ThemolarabsorptivityandSandell'ssensitivityofthemethodsforHg(II)arefoundtobe0.
41x104Lmol-1cm-1and0.
0875g/cm2respectively.
Thespe-cificabsorptivityofthesystemis0.
01142ml/g-1cm-1Hg(II).
Therelativestandarddeviationfortenrepli-cateanalysisofHg(II)is0.
082percent.
Job'sandMolarratiomethodsgavethecompositionoftheHg(II)complexesas1:2(M:L).
thestabilityconstantsofHg(II)complexcalculatedbyJob'smethodisfoundtobe1.
5x1010.
Determinationofmercuryinvariouswater,soilandbiologicalsamplesThepresentmethodisappliedfordeterminationofmercuryin(i)Watersamples,(ii)Soilsamples,and(iii)Biologicalsamples.
Watersamples[4,9]Eachfiltered(withwhattmanNo.
40)watersample[4,9](250ml)wasmixedwith10mlofconcen-tratednitricacidina500mldistillationflask.
Thesamplewasdigestedinthepresenceofanexcesspotassiumpermanganatesolutionaccordingtothemethodrec-ommendedbyFifieldetal[5].
ThesolutionwascooledandneutralizedwithdiluteNH4OHsolution.
Thedigestwastransferredintoa25-mlcalibratedflaskanddi-luteduptothemarkwithdeionizedwater.
TheresultsweregiveninTABLE2.
Soilssamples[5,9]andBiologicalsamples[6,9]Driedfishsamples[6,9]andvarioussoil[5,9]samples2-5gramsweretakenina250mlbeaker.
A6mlofconcentratednitricacidwasaddedandgentlyheatedforhalf-an-hour.
Afterthedisappearanceofthefroth,6mlof1:1nitricacidandperchloricacidwereadded[8-9].
Thecontentsweredigestedfor1hourandrepeatedlytreatedwith6mlportionsofnitricacidandpercholoricacidmixtureuntilthesolutionbecomescolourless.
Theacidicsolutionwasevaporatedtodrynessandthere-sultingwhiteresiduewasdissolvedinminimumvolumeproton,-NH(imino)groupsofhydrazonerespectively.
MassspectrumofAFINHshowsmolecularionpeakatm/z252correspondingtothemolecularionpeakassociatedwithNa(Basepeak).
Otherpeaksduetolossofmethylradicalandfuranylradicalarealsoobservedinmassspectrum.
ThepKavaluesofAFINHThepKavaluesofAFINHweredeterminedbyrecordingtheUV-Visiblespectraofmicromolar(4x10-6M)solutionofthereagentatvariouspHvaluesandbytakingthearithmeticmeansofthevaluesob-tainedfromthemeasurementsatdifferentwavelengthsdeterminedspectrophotometricallyusingPhillipandMerrittmethod[7].
ThevaluesofdeprotonationofAFINHare3.
6(pK1)and6.
3(pK2)correspondingtotheformationofenolformandconjugatedmonoanionformrespectively.
RESULTSANDDISCUSSIONThereagent2-acetylfuranisonicotinoylhydrazoneiseasilypreparedunderrefluxconditions.
A0.
001MsolutionofAFINHisstableformorethantwohours.
Inbuffermedium(pH5.
5),theligandpresumablyex-istsinenolicformandcoordinatesthedivalentmetalionasmonoanion.
Thereagentgivesintensecolourreactiononlywithmercuryandshowmaximumabsor-banceat355nm.
Thereagent(AFINH)isconsideredaspotentialreagentforselectivespectrophotometricdeterminationofmercury(II).
XCCH3NNHCONX=OFigure1:StructureofAFINH.
84Determinationofmercurylevelsinenvironmentalsamplesusing2-acetylfuranFullPaperACAIJ,13(3)2013K.
HussainReddyetal.
85FullPaperACAIJ,13(3)2013AnIndianJournalAnalyticalCHEMISTRYAnalyticalCHEMISTRYThepresentmethod(AFINH)wasappliedforthedeterminationofHg(II)whenpresentaloneandpresentinwater,biologicalandsoilsamplesandresultswerecomparedwithdithizonemethod[10].
APPLICATIONSMercurywasestimatedinvariouswatersamples,biologicalandsoilsamplesbyemployingthepresentmethod.
TheresultsarepresentedinTABLE2CONCLUSIONSThepresentstudyclearlyindicatesthatthemer-curycontentcanbedeterminedinultratracelevelinwater,biologicalandsoilsamplesusingpresentmethod.
Allthesefindingscausegreatconcernregardingpublichealthdemandinganaccuratedeterminationofthismetalionanditmayprovideawarenessamongthepublic.
TABLE3:DeterminationofmercuryinsoilsamplesAmountofmercuryafound(g/ml)NameofthesamplesAFINHmethodDithizonemethodUrbansoil0.
980.
98Agriculturalsoil0.
450.
41Roadsidesoil1.
681.
62aAverageofthreedeterminations.
TABLE4:DeterminationofmercuryinbiologicalsamplesAmountofmercuryafound(g/mlofdriedliver)LiversamplesAINHHmethodDithizonemethodFishliver1.
280.
84Sheepliver0.
830.
86aAverageoffivedeterminationsACKNOWLEDGEMENTTheAuthorSaleemisgratefultoUGCSEROHyderabadForthefinancialassistance,AuthoralsothankDrB.
VSubbaReddyIICTHyderabadandDr.
A.
RathanPrasad,Scientists,forprovidingIR,NMRandMassspectraldata.
REFERENCES[1]M.
HumairaKhan,JamaluddineAhmed;AnalyticalScience,21,507-512(2005).
[2]A.
I.
Vogel;ATextBookofQuantitativeInorganicAnalysis,3rdEdition;ELBSandLongman,325(1975).
[3]G.
Pavlogeorgaters,V.
Kikilias;GlobaNest,Int.
J.
,4(2-3),107-125(2002).
[4]S.
Hamads,S.
Motomizu,K.
Toei;Analyst,113,945-948(1988).
[5]D.
D.
Ferrin,D.
Boyd;BuffersforpHandmetaloncontrol,ChapmanandHall,London,9,128(1974).
[6]F.
W.
Fifield,P.
J.
Haines,(Eds);EnvironmentalAna-lyticalChemistry,BlackwellScience,378(2000).
[7]M.
Kamburova;Talanta,40(5),719-723(1993).
[8]K.
HussainReddy;Ph.
D.
thesis,S.
K.
University,(1983).
[9]KazumiInagakietal.
;TheAnalyst,125,191-196(2000).
[10]F.
D.
Snell,C.
T.
Snell;Colorimetricmethodsofanaly-sis,3rdEdition,11,92(1949).
[11]Z.
Marczenko;Spectrophotometricdeterminationofelements,Wiley,NewYork,241,351,602(1976).
[12]K.
B.
Chandrasekhar,K.
HussainReddy;Indian.
J.
Ch-em.
Sect.
A.
,40,727(2001).
Anexampleforacutemercurypoisoningis"Mina-matadiseases"whichcausesmentaldisturbance,alossofbalance,speech,sightandhearing,difficultyinswallowingandfinallycomaanddeath[2].
Thetoxicityofmercuryde-pendsonitschemicalnature.
Inorganicmercuryhasaveryhighaffinityforproteinsulf-hydrylgroups,accu-mulatesinkidneys,whereasorganicmercuryismoreV.
SaleemBasha1,S.
VidyasagarBabu2,G.
Narasimha3,K.
HussainReddy2*1GovernmentDegreeCollege(Men),Anantapur,A.
P,(INDIA)2DepartmentofChemistry,SriKrishnadevarayaUniversity,Anantapur–515003.
A.
P,(INDIA)3AppliedMicrobiologylaboratory,DepartmentofVirology,SriVenkateswaraUniversity,Tirupati-517502AndhraPradesh,(INDIA)E-mail:khussainreddy@yahoo.
co.
in.
comtoxicsinceitissolubleinfat,thelipidfractionsofmem-branes,andbraintissue.
Mercurytoxicityiscausedmainlybythefactthatitentersthelivingorganismandreactswithdifferentenzymesinhibitingthecatalysisofbasicmetabolicreactions[3].
Theabilityoflivingorgan-ismstoconvertinorganicmercurytoorganicmercurycompoundswhicharemoretoxicandaccumulatetoagreaterextentinsensitivetissues,isalsoaconsiderablefact.
Themainsourcesofmercuryfromwhichitaccu-mulatesintheenvironmentaresyntheticadhesives,airconditionerfilters,amalgams,autoexhausts,anatomicpreservatives,antifoulingpaints,dentalamalgams,bat-teries,cathodetubes,bloodbanksaline,cinnabaretc.
KEYWORDSBiological;Soil;Environmentalsamples;Mercury(II);Substitutedhydrazones;Spectrophotometry.
ABSTRACT2-acetylfuranisonicotinoylhydrazone(AFINH)hasbeensynthesizedandproposedasanewchromogenicreagentforarapid,simple,selective,directandnon-extractivespectrophotometricmethodisdevelopedforthedeterminationofmercury(II)inaqueousdimethylformamide(DMF).
Thereagentgivesdeepyellowcoloured,1:1(M:L)complexwithmercury(II)insodiumacetate-aceticacidbuffermediumofpH5.
5atmax365nm.
Thecolourreactionisinstantaneousandtheabsorbanceremainsconstantforabout12hrs.
Themolarabsorptivityofmercurycomplexis2.
3x103Lmol-1cm-1andsandell'ssensitivityarefoundtobe0.
0888gcm-2.
Themethodwassuccessfullyappliedtoanumberofenvironmental,biologicalandsoilsamples.
Theresultswerecomparablewiththoseobtainedbydithizonemethod.
Theproposedsystemproducedsatisfactoryresultsforthedeter-minationofHg(II).
Theresultsarequiteencouragingandcanbringaware-nessamongthepublic.
2013TradeScienceInc.
-INDIAACAIJ,13(3)2013[81-85]AnIndianJournalVolume13Issue3AnalyticalCHEMISTRYAnalyticalCHEMISTRYISSN:0974-7419.
82Determinationofmercurylevelsinenvironmentalsamplesusing2-acetylfuranFullPaperACAIJ,13(3)2013K.
HussainReddyetal.
83FullPaperACAIJ,13(3)2013AnIndianJournalAnalyticalCHEMISTRYAnalyticalCHEMISTRYDeterminationofmercury(II)Mercury(II)reactswithAFINHinacidicpH(5.
5)togivecolouredcomplexes.
Thecolourreactionisin-stantaneousevenatroomtemperature.
Theorderofadditionofreagent,metalion,buffer,2.
5mlofDMF.
Theabsorbanceofthecolouredcomplexremainscon-stantformorethan2hours.
A10-foldmolarexcessofthereagentisadequateforfullcolourdevelopment.
Ad-ditionofexcessofreagenthasnoadverseeffectontheabsorbanceofthecomplexes.
ThesystemobeysBeer'slawintheconcentrationrange3.
21-32.
1g/mlofmer-cury.
ThemolarabsorptivityandSandell'ssensitivityofthemethodsforHg(II)arefoundtobe0.
41x104Lmol-1cm-1and0.
0875g/cm2respectively.
Thespe-cificabsorptivityofthesystemis0.
01142ml/g-1cm-1Hg(II).
Therelativestandarddeviationfortenrepli-cateanalysisofHg(II)is0.
082percent.
Job'sandMolarratiomethodsgavethecompositionoftheHg(II)complexesas1:2(M:L).
thestabilityconstantsofHg(II)complexcalculatedbyJob'smethodisfoundtobe1.
5x1010.
Determinationofmercuryinvariouswater,soilandbiologicalsamplesThepresentmethodisappliedfordeterminationofmercuryin(i)Watersamples,(ii)Soilsamples,and(iii)Biologicalsamples.
Watersamples[4,9]Eachfiltered(withwhattmanNo.
40)watersample[4,9](250ml)wasmixedwith10mlofconcen-tratednitricacidina500mldistillationflask.
Thesamplewasdigestedinthepresenceofanexcesspotassiumpermanganatesolutionaccordingtothemethodrec-ommendedbyFifieldetal[5].
ThesolutionwascooledandneutralizedwithdiluteNH4OHsolution.
Thedigestwastransferredintoa25-mlcalibratedflaskanddi-luteduptothemarkwithdeionizedwater.
TheresultsweregiveninTABLE2.
Soilssamples[5,9]andBiologicalsamples[6,9]Driedfishsamples[6,9]andvarioussoil[5,9]samples2-5gramsweretakenina250mlbeaker.
A6mlofconcentratednitricacidwasaddedandgentlyheatedforhalf-an-hour.
Afterthedisappearanceofthefroth,6mlof1:1nitricacidandperchloricacidwereadded[8-9].
Thecontentsweredigestedfor1hourandrepeatedlytreatedwith6mlportionsofnitricacidandpercholoricacidmixtureuntilthesolutionbecomescolourless.
Theacidicsolutionwasevaporatedtodrynessandthere-sultingwhiteresiduewasdissolvedinminimumvolumeproton,-NH(imino)groupsofhydrazonerespectively.
MassspectrumofAFINHshowsmolecularionpeakatm/z252correspondingtothemolecularionpeakassociatedwithNa(Basepeak).
Otherpeaksduetolossofmethylradicalandfuranylradicalarealsoobservedinmassspectrum.
ThepKavaluesofAFINHThepKavaluesofAFINHweredeterminedbyrecordingtheUV-Visiblespectraofmicromolar(4x10-6M)solutionofthereagentatvariouspHvaluesandbytakingthearithmeticmeansofthevaluesob-tainedfromthemeasurementsatdifferentwavelengthsdeterminedspectrophotometricallyusingPhillipandMerrittmethod[7].
ThevaluesofdeprotonationofAFINHare3.
6(pK1)and6.
3(pK2)correspondingtotheformationofenolformandconjugatedmonoanionformrespectively.
RESULTSANDDISCUSSIONThereagent2-acetylfuranisonicotinoylhydrazoneiseasilypreparedunderrefluxconditions.
A0.
001MsolutionofAFINHisstableformorethantwohours.
Inbuffermedium(pH5.
5),theligandpresumablyex-istsinenolicformandcoordinatesthedivalentmetalionasmonoanion.
Thereagentgivesintensecolourreactiononlywithmercuryandshowmaximumabsor-banceat355nm.
Thereagent(AFINH)isconsideredaspotentialreagentforselectivespectrophotometricdeterminationofmercury(II).
XCCH3NNHCONX=OFigure1:StructureofAFINH.
84Determinationofmercurylevelsinenvironmentalsamplesusing2-acetylfuranFullPaperACAIJ,13(3)2013K.
HussainReddyetal.
85FullPaperACAIJ,13(3)2013AnIndianJournalAnalyticalCHEMISTRYAnalyticalCHEMISTRYThepresentmethod(AFINH)wasappliedforthedeterminationofHg(II)whenpresentaloneandpresentinwater,biologicalandsoilsamplesandresultswerecomparedwithdithizonemethod[10].
APPLICATIONSMercurywasestimatedinvariouswatersamples,biologicalandsoilsamplesbyemployingthepresentmethod.
TheresultsarepresentedinTABLE2CONCLUSIONSThepresentstudyclearlyindicatesthatthemer-curycontentcanbedeterminedinultratracelevelinwater,biologicalandsoilsamplesusingpresentmethod.
Allthesefindingscausegreatconcernregardingpublichealthdemandinganaccuratedeterminationofthismetalionanditmayprovideawarenessamongthepublic.
TABLE3:DeterminationofmercuryinsoilsamplesAmountofmercuryafound(g/ml)NameofthesamplesAFINHmethodDithizonemethodUrbansoil0.
980.
98Agriculturalsoil0.
450.
41Roadsidesoil1.
681.
62aAverageofthreedeterminations.
TABLE4:DeterminationofmercuryinbiologicalsamplesAmountofmercuryafound(g/mlofdriedliver)LiversamplesAINHHmethodDithizonemethodFishliver1.
280.
84Sheepliver0.
830.
86aAverageoffivedeterminationsACKNOWLEDGEMENTTheAuthorSaleemisgratefultoUGCSEROHyderabadForthefinancialassistance,AuthoralsothankDrB.
VSubbaReddyIICTHyderabadandDr.
A.
RathanPrasad,Scientists,forprovidingIR,NMRandMassspectraldata.
REFERENCES[1]M.
HumairaKhan,JamaluddineAhmed;AnalyticalScience,21,507-512(2005).
[2]A.
I.
Vogel;ATextBookofQuantitativeInorganicAnalysis,3rdEdition;ELBSandLongman,325(1975).
[3]G.
Pavlogeorgaters,V.
Kikilias;GlobaNest,Int.
J.
,4(2-3),107-125(2002).
[4]S.
Hamads,S.
Motomizu,K.
Toei;Analyst,113,945-948(1988).
[5]D.
D.
Ferrin,D.
Boyd;BuffersforpHandmetaloncontrol,ChapmanandHall,London,9,128(1974).
[6]F.
W.
Fifield,P.
J.
Haines,(Eds);EnvironmentalAna-lyticalChemistry,BlackwellScience,378(2000).
[7]M.
Kamburova;Talanta,40(5),719-723(1993).
[8]K.
HussainReddy;Ph.
D.
thesis,S.
K.
University,(1983).
[9]KazumiInagakietal.
;TheAnalyst,125,191-196(2000).
[10]F.
D.
Snell,C.
T.
Snell;Colorimetricmethodsofanaly-sis,3rdEdition,11,92(1949).
[11]Z.
Marczenko;Spectrophotometricdeterminationofelements,Wiley,NewYork,241,351,602(1976).
[12]K.
B.
Chandrasekhar,K.
HussainReddy;Indian.
J.
Ch-em.
Sect.
A.
,40,727(2001).
- cm003hh.com相关文档
- 展品003hh.com
- implied003hh.com
- 寄存器003hh.com
- peace003hh.com
- wait003hh.com
- Flowchart003hh.com
博鳌云¥799/月,香港110Mbps(含10M CN2)大带宽独立服务器/E3/8G内存/240G/500G SSD或1T HDD
博鳌云是一家以海外互联网基础业务为主的高新技术企业,运营全球高品质数据中心业务。自2008年开始为用户提供服务,距今11年,在国人商家中来说非常老牌。致力于为中国用户提供域名注册(国外接口)、免费虚拟主机、香港虚拟主机、VPS云主机和香港、台湾、马来西亚等地服务器租用服务,各类网络应用解決方案等领域的专业网络数据服务。商家支持支付宝、微信、银行转账等付款方式。目前香港有一款特价独立服务器正在促销,...

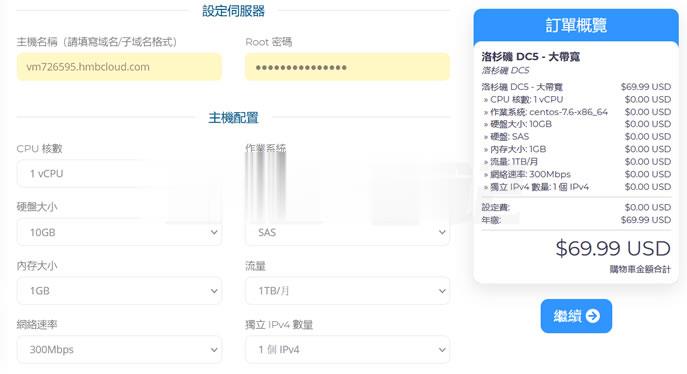
半月湾hmbcloud升级500Mbps带宽,原生VPS,$4.99/月
关于半月湾HMBCloud商家之前也有几篇那文章介绍过这个商家的产品,对于他们家的其他产品我都没有多加留意,而是对他们家的DC5机房很多人还是比较喜欢的,这个比我们有些比较熟悉的某商家DC6 DC9机房限时,而且半月湾HMBCloud商家是相对便宜的。关于半月湾DC5机房的方案选择和介绍:1、半月湾三网洛杉矶DC5 CN2 GIA同款DC6 DC9 1G内存 1TB流量 月$4.992、亲测选择半...
美得云(15元/月)美国cera 2核4G 15元/月 香港1核 1G 3M独享
美得云怎么样?美得云好不好?美得云是第一次来推广软文,老板人脾气特别好,能感觉出来会用心对待用户。美得云这次为大家提供了几款性价比十分高的产品,美国cera 2核4G 15元/月 香港1核 1G 3M独享 15元/月,并且还提供了免费空间给大家使用。嘻嘻 我也打算去白嫖一个空间了。新用户注册福利-8折优惠码:H2dmBKbF 截止2021.10.1结束。KVM架构,99.99%高可用性,依托BGP...
003hh.com为你推荐
-
特朗普取消访问丹麦特朗普当选总统后对准备出国留学的学生有什么影响中老铁路中国有哪些正在修的铁路百度商城百度知道一般一天能挣多少钱?阿丽克丝·布莱肯瑞吉行尸走肉第六季女演员mathplayer如何学好理科关键字关键词标签里写多少个关键词为最好比肩工场命比肩多 是什么意思啊?www.7160.com电影网站有那些罗伦佐娜维洛娜毛周角化修复液治疗毛周角化有用吗?谁用过?能告诉我吗?月神谭求男变女类的变身小说