http://www.aqsiqccc.com/en/aqsiq/sfda-9.html
www.585ccc.com 时间:2021-03-19 阅读:()
LocationHome>ServiceProject>ApplicationandEvaluationofnewcosmeticingredientsguideDate2011-05-12CosmeticsrawmaterialsontheissuanceofnewguidelinesforthereviewofthenotificationandreportingStateFoodandDrugAdministrationXu[2011]No.
207May12,2011releaseProvinces,autonomousregionsandmunicipalitiesFoodandDrugAdministration(DrugAdministration),theunit:Newcosmeticingredientstoenhancetheadministrativelicensingworktoensurequalityandsafetyofcosmeticproducts,basedon"cosmeticsHealthSupervision"anditsImplementingRulesandotherrelevantprovisionsoftheStateFoodandDrugAdministrationtodevelopa"declarationofnewcosmeticrawmaterialsandguidelinesforthereview.
"Isherebyissued,pleasecomply.
ApplicationandEvaluationofnewcosmeticingredientsguideStateFoodandDrugAdministrationMay12,2011Attachment:ApplicationandEvaluationofnewcosmeticingredientsguideThisguideisintendedtoguidethedeclarationofnewcosmeticingredientsandreview.
First,thedefinitionofnewcosmeticingredientsNewcosmeticrawmaterialisusedincosmeticsinthecountryforthefirsttimetheproductionofnaturalorartificialmaterials.
Second,thesecurityrequirementsofnewcosmeticingredientsNewcosmeticingredientsinthenormalandreasonableforeseeableconditionsofuseshallnotcauseharmtohumanhealth.
Toxicologicalevaluationofnewinformationoncosmeticingredientsshouldincludetoxicologicalsafetyevaluationreview,thenecessarytoxicologytestinginformationsecurityrisksandpossiblematerialsforsafetyassessmentdata.
Cosmeticsrawmaterialsaregenerallyinneedofnewtoxicologytestsfollowing:(A)acuteoralandacutedermaltoxicitytest;(B)theskinandacuteeyeirritation/corrosiontest;(C)theskinallergytest;(D)andlight-sensitiveskinphototoxicitytest(UVabsorptionpropertiesofrawmaterialshastobedonewhenthetrial);(E)mutationtest(shouldatleastincludeagenemutationtestandachromosomeaberrationtest);(F)sub-chronicoralanddermaltoxicitytest;(G)teratogenictest;(H)chronictoxicity/carcinogenicitytestcombination;(Ix)toxicokineticsanddynamicstest;(X)accordingtothecharacteristicsanduseofrawmaterials,butalsoconsiderothernecessarytests.
Ifthenewmaterialhasbeenusedincosmeticsandchemicalstructureandpropertiesofsimilarmaterials,youcanconsiderreducingsomeofthetest.
Thisguideprovidesinformationfortheprinciplerequirementsoftoxicologytests,accordingtothephysicalandchemicalpropertiesofrawmaterials,QSAR,toxicology,clinicalresearch,epidemiologyandpopulationsimilartothetoxicityofcompounds,suchasinformationapilotprojecttoincreaseorreduce.
Third,newcosmeticingredientsadministrativelicensingrequirementsfordisclosureofinformationApplyfornewcosmeticingredientscosmeticadministrativelicenseshallaccepttheadministrativelicensingrequirementstosubmitinformationreturns.
Specificrequirementsareasfollows:(A)newcosmeticrawmaterials,administrativelicensingapplicationform(B)developmentreport1MaterialsR&Dbackground,processandrelatedtechnicalinformation.
(2)thenameofrawmaterial,origin,molecularweight,molecularformula,chemicalstructure,physicalandchemicalproperties.
(1)Name:rawmaterials,includingthechemicalname(IUPACnameand/orCASname),INCInameandtheirChinesetranslation,tradenamesandCASnumbers.
Materialshouldalsoindicatethenameoftheuseofrawmaterialsspecifications.
NaturalrawmaterialsshouldalsoprovidetheLatinscientificname.
(2)Source:rawmaterialsshouldnotbeacompoundformedinthematerialduetounavoidabletechnicalreasons,theexistenceofsolvents,stabilizers,carriers,etc.
,except.
Naturalmaterialsshouldbeasinglesource,andprovidestheuseofpartsandsoon.
Allplantshavebeenallowedtobeusedascosmeticrawmaterials,notallpartsoftheplantmaterialandthenanewdeclaration.
(3)molecularweight,molecularformula,chemicalstructure:chemicalstructureshouldprovidethebasisforconfirmation(eg,NMR,elementalanalysis,massspectrometry,infraredspectra,etc.
)andanalyticalresults,shouldalsoprovidepolymerrelativetotheaveragemolecularweightanditsdistribution.
(4)physicalandchemicalpropertiesinclude:color,odor,state,solubility,meltingpoint,boilingpoint,specificgravity,vaporpressure,pHvalue,pKavalue,refractiveindex,opticalrotationandsoon.
3ingredientsincosmeticsintendeduse,scopeofuseforsecurityandtheuseoflimitedbasis,cautions,warnings,etc.
4ingredientsinaforeigncountry(region)isusedincosmeticscasedescriptions.
(C)theproductionprocessdescriptionanddiagramShouldindicatethenewcosmeticrawmaterialstoproducethemajorstepsinvolvedintheprocess,proceduresandparameters,suchasrawmaterialsshouldbelisted,thereactionconditions(temperature,pressure,etc.
),additives(catalysts,stabilizers),intermediatesandby-productsandthepreparationstepsetc.
;thecaseofnaturalextracts,shouldindicatetheprocessing,extractionmethod,extractionconditions,theuseofsolvents,mayberesidualimpuritiesorsolvent.
(D)rawmaterialqualityandsafetycontrolrequirementsShouldincludespecifications,testmethods,theremaybeasecurityrisksubstancesandcontrolmeasuresandsoon.
1.
Specificationsinclude:purityorcontentofimpurityspeciesandtheirrespectivecontent(residualmonomerandpolymercontenttobedescribed)andotherindicatorsofqualityandsafetycontrol,duetounavoidabletechnicalreasonsexistintherawmaterialsofsolvent,stabilizer,carrier,etc.
typesandtheirrespectivecontent,otherphysicalandchemicalparameters,shelflifeandstorageconditions;thecaseofnaturalplantextracts,shouldbeclearindicatorsofitsqualityandsafetycontrol.
2detectionmethods:qualitativeandquantitativematerialtestingmethods,testingmethodsimpurities.
3possiblesecurityrisksubstancesandtheircontrolmeasures.
(E)toxicologicalsafetyevaluationdata(includingrawmaterials,theremaybesecurityrisksinthesafetyassessmentofmaterialinformation)Toxicologytestdatacanbetheapplicant'stestdata,scientificliteratureanddomesticandforeigngovernmentofficialwebsite,thecontentofwebsitesofinternationalorganizations.
1applyfornewcosmeticingredients,cosmetics,newmaterialsgenerallyshouldberequiredtosubmitsafetydatatoxicologytests.
(2)oneofthefollowingcircumstances,accordingtothefollowingrequirementstosubmittoxicologytestdata.
Accordingtothecharacteristicsanduseofrawmaterials,ifnecessary,mayrequestanincreaseorreductionrelatedtoexperimentaldata.
(1)whodonothavepreservatives,sunscreens,colorantsanddyes,andfunctionalingredientsfromasafetypointofviewnotincludedinthe"HygienicStandardforCosmetics"Restrictedsubstancestablesnewcosmeticrawmaterials,shouldsubmitthefollowinginformation:1)Theacuteoralandacutedermaltoxicitytest;2)skinandacuteeyeirritation/corrosiontest;3)skinallergytest;4)andlight-sensitiveskinphototoxicitytest(UVabsorptionpropertiesofrawmaterialshastobedonewhenthetwotrials);5)mutationtest(shouldatleastincludeagenemutationtestandachromosomeaberrationtest);6)sub-chronicoralordermaltoxicitytest.
Iftherawmaterialsusedincosmetics,thepossibilityoforalintakeoflarge,shouldprovidesub-chronicoraltoxicitytest.
(2)complywiththeconditions(1),andbyforeign(regional)authoritiesoncosmeticingredientscatalogreproducesmorethanfouryears,involvesnoharmtohumanhealthmayberelatedtoliterature,shouldsubmitthefollowinginformation:1)Theacuteoralandacutedermaltoxicitytest;2)skinandacuteeyeirritation/corrosiontest;3)skinallergytest;4)andlight-sensitiveskinphototoxicitytest(UVabsorptionpropertiesofrawmaterialshastobedonewhenthetwotrials);5)mutationtest(shouldatleastincludeagenemutationtestandachromosomeaberrationtest).
(3)Whereahistoryofsafehumanconsumption,suchasdomesticandforeigngovernmentofficialoragencyorauthorityissuedbythesafetyassessmentoffoodingredientsconsideredsafeanditsextracts,theStateCounciladministrativedepartmentannouncedasbothfoodandpharmaceuticalitems,etc.
shouldsubmitthefollowinginformation:1)skinandacuteeyeirritation/corrosiontest;2)Theskinallergytest;3)andlight-sensitiveskinphototoxicitytest(UVabsorptionpropertiesofrawmaterialshastobedonewhenthetrial).
(4)byoneormorestructuralunitsconnectedbycovalentbonds,relativetotheaveragemolecularweightgreaterthan1000daltonsnewpolymersascosmeticrawmaterials,shouldsubmitthefollowinginformation:1)skinandacuteeyeirritation/corrosiontest;2)skinphototoxicitytest(UVabsorptionpropertiesofrawmaterialshastobedonewhenthetrial).
(5)Whereaforeign(regional)authoritiesevaluationconcludedthatitissafetouseincosmeticsofthenewmaterial,reportingisnotrequiredtoprovidetoxicologicaltestdata,butshouldbesubmittedtoforeigncountries(regions)toassesstheconclusionsofevaluationreportsandrelatedinformation.
Foreign(region)approvedanewcosmeticingredients,shouldbesubmittedtotheapprovalcertificate.
(F)importsnewcosmeticrawmaterials,theapplicantshallbesubmittedtotheadministrativelicensinghasbeenfiledtodeclaretheresponsibleunitsinChina,powerofattorneyandacopyoftheirreportingresponsibilitiesontheadministrativelicensingunitinChinabusinesslicenseandofficialseal.
(G)maycontributetotheadministrativelicensingandotherinformation.
Applicantsshouldbebasedonthenewmaterialpropertiesaccordingtotheaboverequestedinformation,unlesstherelevantrequirementsdonotapply.
Attachedasamplesentforscreening.
Fourth,theprincipleofnewcosmeticingredientsreview(A)fortheapplicanttosubmitthesafetyevaluationofnewcosmeticingredientsdataintegrity,reasonableandscientificconductofthereview:1safetyevaluationofinformationcontentiscompleteandconsistentinformationrequirements;2basedonwhetherthescientific,criticaldataisreasonable,analysisislogical,conclusioniscorrect;3keynewsourceofrawmaterialsofcosmeticsreview,physicalandchemicalnature,purpose,scope,andlimitedusebasis,productionprocesses,qualityandsafetycontrolrequirementsandthenecessarytoxicologicalevaluationofinformation.
(B)Thereviewconsideredcosmeticsafetyevaluationofnewrawmaterialproblems,theexpertsshouldreviewtherelevantprovisionsofcosmeticsregulatoryandscientificbasis,tomakespecificcomments.
Theapplicantwithintheprescribedtimelimitshouldprovideappropriatesafetyevaluation.
(C)Withthedevelopmentofscientificresearch,theStateFoodandDrugAdministrationhasapprovedcosmeticscanre-evaluationofnewmaterials.
Fifth,aspecialtypeofreportingandreviewofnewcosmeticrawmaterialrequirementswillbeforthcoming.
VIAbbreviations(A)IUPAC,InternationalUnionofPureandAppliedChemistry(InternationalUnionofPureandAppliedChemistry)abbreviations.
(B)CAS,theAmericanChemicalAbstractsService(ChemicalAbstractsService)abbreviations.
(C)INCI,Internationalcosmeticingredientsnamed(InternationalNomenclatureCosmeticIngredient)abbreviation.
ThisguidebytheStateFoodandDrugAdministrationresponsiblefortheinterpretation.
ThisguideisfromJuly1,2011shallcomeintoforce.
Previouslypublishedreportandreviewofnewcosmeticingredientsregulationsinconsistentwiththisguideinordertoprevailinthisguide.
BeijingTongRuiLianTechnologyCo.
,Ltd.
Tel:86-10-6428460586-10-64132180Fax:86-10-64132180E-mail:bj500@126.
combj2180@gmail.
com
207May12,2011releaseProvinces,autonomousregionsandmunicipalitiesFoodandDrugAdministration(DrugAdministration),theunit:Newcosmeticingredientstoenhancetheadministrativelicensingworktoensurequalityandsafetyofcosmeticproducts,basedon"cosmeticsHealthSupervision"anditsImplementingRulesandotherrelevantprovisionsoftheStateFoodandDrugAdministrationtodevelopa"declarationofnewcosmeticrawmaterialsandguidelinesforthereview.
"Isherebyissued,pleasecomply.
ApplicationandEvaluationofnewcosmeticingredientsguideStateFoodandDrugAdministrationMay12,2011Attachment:ApplicationandEvaluationofnewcosmeticingredientsguideThisguideisintendedtoguidethedeclarationofnewcosmeticingredientsandreview.
First,thedefinitionofnewcosmeticingredientsNewcosmeticrawmaterialisusedincosmeticsinthecountryforthefirsttimetheproductionofnaturalorartificialmaterials.
Second,thesecurityrequirementsofnewcosmeticingredientsNewcosmeticingredientsinthenormalandreasonableforeseeableconditionsofuseshallnotcauseharmtohumanhealth.
Toxicologicalevaluationofnewinformationoncosmeticingredientsshouldincludetoxicologicalsafetyevaluationreview,thenecessarytoxicologytestinginformationsecurityrisksandpossiblematerialsforsafetyassessmentdata.
Cosmeticsrawmaterialsaregenerallyinneedofnewtoxicologytestsfollowing:(A)acuteoralandacutedermaltoxicitytest;(B)theskinandacuteeyeirritation/corrosiontest;(C)theskinallergytest;(D)andlight-sensitiveskinphototoxicitytest(UVabsorptionpropertiesofrawmaterialshastobedonewhenthetrial);(E)mutationtest(shouldatleastincludeagenemutationtestandachromosomeaberrationtest);(F)sub-chronicoralanddermaltoxicitytest;(G)teratogenictest;(H)chronictoxicity/carcinogenicitytestcombination;(Ix)toxicokineticsanddynamicstest;(X)accordingtothecharacteristicsanduseofrawmaterials,butalsoconsiderothernecessarytests.
Ifthenewmaterialhasbeenusedincosmeticsandchemicalstructureandpropertiesofsimilarmaterials,youcanconsiderreducingsomeofthetest.
Thisguideprovidesinformationfortheprinciplerequirementsoftoxicologytests,accordingtothephysicalandchemicalpropertiesofrawmaterials,QSAR,toxicology,clinicalresearch,epidemiologyandpopulationsimilartothetoxicityofcompounds,suchasinformationapilotprojecttoincreaseorreduce.
Third,newcosmeticingredientsadministrativelicensingrequirementsfordisclosureofinformationApplyfornewcosmeticingredientscosmeticadministrativelicenseshallaccepttheadministrativelicensingrequirementstosubmitinformationreturns.
Specificrequirementsareasfollows:(A)newcosmeticrawmaterials,administrativelicensingapplicationform(B)developmentreport1MaterialsR&Dbackground,processandrelatedtechnicalinformation.
(2)thenameofrawmaterial,origin,molecularweight,molecularformula,chemicalstructure,physicalandchemicalproperties.
(1)Name:rawmaterials,includingthechemicalname(IUPACnameand/orCASname),INCInameandtheirChinesetranslation,tradenamesandCASnumbers.
Materialshouldalsoindicatethenameoftheuseofrawmaterialsspecifications.
NaturalrawmaterialsshouldalsoprovidetheLatinscientificname.
(2)Source:rawmaterialsshouldnotbeacompoundformedinthematerialduetounavoidabletechnicalreasons,theexistenceofsolvents,stabilizers,carriers,etc.
,except.
Naturalmaterialsshouldbeasinglesource,andprovidestheuseofpartsandsoon.
Allplantshavebeenallowedtobeusedascosmeticrawmaterials,notallpartsoftheplantmaterialandthenanewdeclaration.
(3)molecularweight,molecularformula,chemicalstructure:chemicalstructureshouldprovidethebasisforconfirmation(eg,NMR,elementalanalysis,massspectrometry,infraredspectra,etc.
)andanalyticalresults,shouldalsoprovidepolymerrelativetotheaveragemolecularweightanditsdistribution.
(4)physicalandchemicalpropertiesinclude:color,odor,state,solubility,meltingpoint,boilingpoint,specificgravity,vaporpressure,pHvalue,pKavalue,refractiveindex,opticalrotationandsoon.
3ingredientsincosmeticsintendeduse,scopeofuseforsecurityandtheuseoflimitedbasis,cautions,warnings,etc.
4ingredientsinaforeigncountry(region)isusedincosmeticscasedescriptions.
(C)theproductionprocessdescriptionanddiagramShouldindicatethenewcosmeticrawmaterialstoproducethemajorstepsinvolvedintheprocess,proceduresandparameters,suchasrawmaterialsshouldbelisted,thereactionconditions(temperature,pressure,etc.
),additives(catalysts,stabilizers),intermediatesandby-productsandthepreparationstepsetc.
;thecaseofnaturalextracts,shouldindicatetheprocessing,extractionmethod,extractionconditions,theuseofsolvents,mayberesidualimpuritiesorsolvent.
(D)rawmaterialqualityandsafetycontrolrequirementsShouldincludespecifications,testmethods,theremaybeasecurityrisksubstancesandcontrolmeasuresandsoon.
1.
Specificationsinclude:purityorcontentofimpurityspeciesandtheirrespectivecontent(residualmonomerandpolymercontenttobedescribed)andotherindicatorsofqualityandsafetycontrol,duetounavoidabletechnicalreasonsexistintherawmaterialsofsolvent,stabilizer,carrier,etc.
typesandtheirrespectivecontent,otherphysicalandchemicalparameters,shelflifeandstorageconditions;thecaseofnaturalplantextracts,shouldbeclearindicatorsofitsqualityandsafetycontrol.
2detectionmethods:qualitativeandquantitativematerialtestingmethods,testingmethodsimpurities.
3possiblesecurityrisksubstancesandtheircontrolmeasures.
(E)toxicologicalsafetyevaluationdata(includingrawmaterials,theremaybesecurityrisksinthesafetyassessmentofmaterialinformation)Toxicologytestdatacanbetheapplicant'stestdata,scientificliteratureanddomesticandforeigngovernmentofficialwebsite,thecontentofwebsitesofinternationalorganizations.
1applyfornewcosmeticingredients,cosmetics,newmaterialsgenerallyshouldberequiredtosubmitsafetydatatoxicologytests.
(2)oneofthefollowingcircumstances,accordingtothefollowingrequirementstosubmittoxicologytestdata.
Accordingtothecharacteristicsanduseofrawmaterials,ifnecessary,mayrequestanincreaseorreductionrelatedtoexperimentaldata.
(1)whodonothavepreservatives,sunscreens,colorantsanddyes,andfunctionalingredientsfromasafetypointofviewnotincludedinthe"HygienicStandardforCosmetics"Restrictedsubstancestablesnewcosmeticrawmaterials,shouldsubmitthefollowinginformation:1)Theacuteoralandacutedermaltoxicitytest;2)skinandacuteeyeirritation/corrosiontest;3)skinallergytest;4)andlight-sensitiveskinphototoxicitytest(UVabsorptionpropertiesofrawmaterialshastobedonewhenthetwotrials);5)mutationtest(shouldatleastincludeagenemutationtestandachromosomeaberrationtest);6)sub-chronicoralordermaltoxicitytest.
Iftherawmaterialsusedincosmetics,thepossibilityoforalintakeoflarge,shouldprovidesub-chronicoraltoxicitytest.
(2)complywiththeconditions(1),andbyforeign(regional)authoritiesoncosmeticingredientscatalogreproducesmorethanfouryears,involvesnoharmtohumanhealthmayberelatedtoliterature,shouldsubmitthefollowinginformation:1)Theacuteoralandacutedermaltoxicitytest;2)skinandacuteeyeirritation/corrosiontest;3)skinallergytest;4)andlight-sensitiveskinphototoxicitytest(UVabsorptionpropertiesofrawmaterialshastobedonewhenthetwotrials);5)mutationtest(shouldatleastincludeagenemutationtestandachromosomeaberrationtest).
(3)Whereahistoryofsafehumanconsumption,suchasdomesticandforeigngovernmentofficialoragencyorauthorityissuedbythesafetyassessmentoffoodingredientsconsideredsafeanditsextracts,theStateCounciladministrativedepartmentannouncedasbothfoodandpharmaceuticalitems,etc.
shouldsubmitthefollowinginformation:1)skinandacuteeyeirritation/corrosiontest;2)Theskinallergytest;3)andlight-sensitiveskinphototoxicitytest(UVabsorptionpropertiesofrawmaterialshastobedonewhenthetrial).
(4)byoneormorestructuralunitsconnectedbycovalentbonds,relativetotheaveragemolecularweightgreaterthan1000daltonsnewpolymersascosmeticrawmaterials,shouldsubmitthefollowinginformation:1)skinandacuteeyeirritation/corrosiontest;2)skinphototoxicitytest(UVabsorptionpropertiesofrawmaterialshastobedonewhenthetrial).
(5)Whereaforeign(regional)authoritiesevaluationconcludedthatitissafetouseincosmeticsofthenewmaterial,reportingisnotrequiredtoprovidetoxicologicaltestdata,butshouldbesubmittedtoforeigncountries(regions)toassesstheconclusionsofevaluationreportsandrelatedinformation.
Foreign(region)approvedanewcosmeticingredients,shouldbesubmittedtotheapprovalcertificate.
(F)importsnewcosmeticrawmaterials,theapplicantshallbesubmittedtotheadministrativelicensinghasbeenfiledtodeclaretheresponsibleunitsinChina,powerofattorneyandacopyoftheirreportingresponsibilitiesontheadministrativelicensingunitinChinabusinesslicenseandofficialseal.
(G)maycontributetotheadministrativelicensingandotherinformation.
Applicantsshouldbebasedonthenewmaterialpropertiesaccordingtotheaboverequestedinformation,unlesstherelevantrequirementsdonotapply.
Attachedasamplesentforscreening.
Fourth,theprincipleofnewcosmeticingredientsreview(A)fortheapplicanttosubmitthesafetyevaluationofnewcosmeticingredientsdataintegrity,reasonableandscientificconductofthereview:1safetyevaluationofinformationcontentiscompleteandconsistentinformationrequirements;2basedonwhetherthescientific,criticaldataisreasonable,analysisislogical,conclusioniscorrect;3keynewsourceofrawmaterialsofcosmeticsreview,physicalandchemicalnature,purpose,scope,andlimitedusebasis,productionprocesses,qualityandsafetycontrolrequirementsandthenecessarytoxicologicalevaluationofinformation.
(B)Thereviewconsideredcosmeticsafetyevaluationofnewrawmaterialproblems,theexpertsshouldreviewtherelevantprovisionsofcosmeticsregulatoryandscientificbasis,tomakespecificcomments.
Theapplicantwithintheprescribedtimelimitshouldprovideappropriatesafetyevaluation.
(C)Withthedevelopmentofscientificresearch,theStateFoodandDrugAdministrationhasapprovedcosmeticscanre-evaluationofnewmaterials.
Fifth,aspecialtypeofreportingandreviewofnewcosmeticrawmaterialrequirementswillbeforthcoming.
VIAbbreviations(A)IUPAC,InternationalUnionofPureandAppliedChemistry(InternationalUnionofPureandAppliedChemistry)abbreviations.
(B)CAS,theAmericanChemicalAbstractsService(ChemicalAbstractsService)abbreviations.
(C)INCI,Internationalcosmeticingredientsnamed(InternationalNomenclatureCosmeticIngredient)abbreviation.
ThisguidebytheStateFoodandDrugAdministrationresponsiblefortheinterpretation.
ThisguideisfromJuly1,2011shallcomeintoforce.
Previouslypublishedreportandreviewofnewcosmeticingredientsregulationsinconsistentwiththisguideinordertoprevailinthisguide.
BeijingTongRuiLianTechnologyCo.
,Ltd.
Tel:86-10-6428460586-10-64132180Fax:86-10-64132180E-mail:bj500@126.
combj2180@gmail.
com
- http://www.aqsiqccc.com/en/aqsiq/sfda-9.html相关文档
- tableswww.585ccc.com
- contactedwww.585ccc.com
- releasedwww.585ccc.com
- outstandingwww.585ccc.com
- Swanwww
- www.585ccc.com手机ccc认证查询,求网址
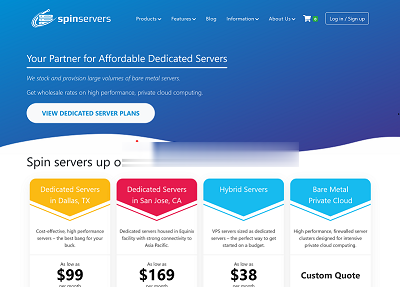
spinservers:圣何塞物理机7.5折,$111/月,2*e5-2630Lv3/64G内存/2T SSD/10Gbps带宽
spinservers美国圣何塞机房的独立服务器补货120台,默认接入10Gbps带宽,给你超高配置,这价格目前来看好像真的是无敌手,而且可以做到下单后30分钟内交货,都是预先部署好了的。每一台机器用户都可以在后台自行安装、重装、重启、关机操作,无需人工参与! 官方网站:https://www.spinservers.com 比特币、信用卡、PayPal、支付宝、webmoney、Payssi...
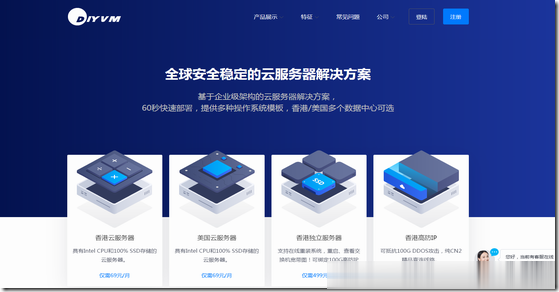
DiyVM:2G内存/50G硬盘/元起线路香港vps带宽CN2线路,香港VPS五折月付50元起
DiyVM是一家低调国人VPS主机商,成立于2009年,提供的产品包括VPS主机和独立服务器租用等,数据中心包括香港沙田、美国洛杉矶、日本大阪等,VPS主机基于XEN架构,均为国内直连线路,主机支持异地备份与自定义镜像,可提供内网IP。最近,商家对香港机房VPS提供5折优惠码,最低2GB内存起优惠后仅需50元/月。下面就以香港机房为例,分享几款VPS主机配置信息。CPU:2cores内存:2GB硬...
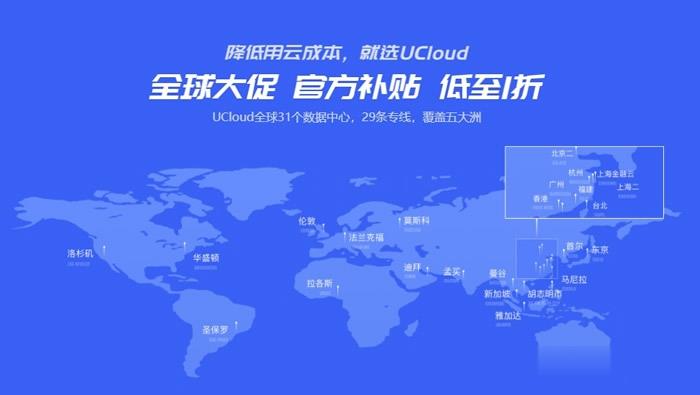
UCloud:美国云服务器,洛杉矶节点大促,低至7元起/1个月
ucloud美国云服务器怎么样?ucloud是国内知名云计算品牌服务商家,目前推出全球多地机房的海外云服务器。UCloud主打的优势是海外多机房,目前正在进行的2021全球大促活动参与促销的云服务器机房就多达18个。UCloud新一代旗舰产品快杰云服务器已上线洛杉矶节点,覆盖北美和亚太地区,火热促销中, 首月低至7元,轻松体验具备优秀性能与极高性价比的快杰云服务器。点击进入:ucloud美国洛杉矶...
www.585ccc.com为你推荐
-
7788k.comwww.k6320.com 大家给我看看这网站是真是假...rawtoolsU盘显示是RAW格式怎么办罗伦佐娜维洛娜毛周角化修复液治疗毛周角化有用吗?谁用过?能告诉我吗?www.622hh.comwww.710av.com怎么不可以看了www.7788dy.comwww.tom365.com这个免费的电影网站有毒吗?ip查询器怎么样查看自己电脑上的IP地址www.bbb551.com100bbb网站怎样上不去了ww.66bobo.comfq55点com是什么网站yinrentangWeichentang正品怎么样,谁知道?haole012.com012.com网站真的可以挂Q升级吗?