Antifibrogenicm.kan84.net
m.kan84.net 时间:2021-03-19 阅读:()
WorldJTraditChinMed|Volume3|Issue3|JulySeptember201746ReviewArticleIntroductionXiaoChaiHuTang(XCHT,MinorBupleurumDecoction),alsoknownasShosaikotoinJapan,isapopularChineseherbalformulaappliedinAsia,containingsevenmedicinalplants:bupleurumroot,pinelliaternata,scutellariaroot,jujubaefructus,panaxginseng,licorice,andginger.
Table1liststheprincipalconstituentsofeachherbcomponent.
ThisformulaisderivedfromthebookTreatiseonColdDamageDiseases,writtenbyZhangZhongjing(afamousphysicianofthelateEasternHanDynasty,about200A.
D.
),andnow,manyexperimentalandclinicalresearchesincludingprospective,randomized,andplacebocontrolledtrialshaveinvolvedtheclinicefficacyofXCHT.
Althoughithasbeenwidelyusedbypatientswithdifferentkindsofhepaticdiseases,suchashepatitisandliverfibrosis,itsbenefitsclaimedneedanaccurateassessment.
ThisreviewsumsuptheavailablescientificfindingsaboutXCHTandhepaticdiseases,pointingoutthedirectionforfurtherstudies.
MechanismsofActionHepatoprotectiveeffectsXCHThelpstomaintainthebalanceamongtheimmunologicalcytokines,whichmayhaveimmunologicalbenefitstochronicviralhepatitis.
[1]Forexample,scutellariarootandlicoriceinXCHTcouldinducetheproductionofcytokinessuchasinterleukin(IL)1β,tumornecrosisfactor(TNF)α,andgranulocytecolonystimulatingfactor(GCSF)frommonocytes/macrophages.
[2]Kakumuetal.
[3]foundXCHTincreasedinterferonγ(IFNγ)andantibodyagainstHBV,strengtheningcellularandhumoralimmuneresponses.
ToXiaoChaiHuTangforLiverDiseases:ALiteratureReviewYiWanga,LiLib,YanMeiChenga,ShengLiangZhuaaDepartmentofGastroenterology,YueyangHospitalofIntegratedTraditionalChineseandWesternMedicine,ShanghaiUniversityofTraditionalChineseMedicine,bClinicalResearchInstituteofIntegratedTraditionalChineseandWesternMedicine,YueyangHospitalofIntegratedTraditionalChineseandWesternMedicine,ShanghaiUniversityofTraditionalChineseMedicine,Shanghai200437,ChinaTheobjectiveofthisstudyistosummarizethepharmacologicaleffectsandthemechanismsofactionofXiaoChaiHuTang(XCHT,MinorBupleurumDecoction)onliverdiseases,soastogiverelevantresearchersavaluableinsightandbenefitpatientswithhepatopathy.
PubMedwasusedtosearchforandcollectscientificpublicationsrelatedtoXCHTandliverdiseasesfrom1986to2016.
Theavailablescientificresultsorevidencewereread,classified,andanalyzed.
XCHTshowedclinicalefficacyinpatientswithhepaticdiseasesincludinghepatitis,hepaticfibrosis,andhepatoma.
Themechanismsinvolvedtheproductionofcytokines,theregulationofimmunefunction,thesuppressionoflipidperoxidation,etc.
,XCHTmightworkonthemetabolismofsomemedicationssuchastolbutamidebytheregulationofgastricemptyingandintragastricpH.
XCHTexhibitedaverylowtoxicityprofile,suchasinterstitialpneumoniaduetodurationofmedication,patients'age,anddrugcombination.
XCHThasbeenaeutherapeuticsupplementalremedyforliverdiseases.
However,manymechanismsofactionandeffectsofXCHTonnewtypesofliverdiseasesstillremainunclear,somoreandmoreanimalexperimentsandhumanclinicaltrialsareneededtoobtainenoughproofsfortheclinicaluseofXCHTinnewtypesofhepatosissuchasnonalcoholicfattyliverdiseaseandautoimmuneliverdisease.
Keywords:Hepaticfibrosis,hepatitis,hepatoma,interstitialpneumonia,tolbutamide,XiaoChaiHuTang(minorbupleurumdecoction)AccessthisarticleonlineQuickResponseCode:Website:www.
wjtcm.
netDOI:10.
4103/wjtcm.
wjtcm_4_17AbstractDownloadfreefromwww.
wjtcm.
netAddressforcorrespondence:Prof.
ShengLiangZhu,DepartmentofGastroenterology,YueyangHospitalofIntegratedTraditionalChineseandWesternMedicine,ShanghaiUniversityofTraditionalChineseMedicine,Shanghai200437,China.
Email:zhushengliang999@126.
comHowtocitethisarticle:WangY,LiL,ChengYM,ZhuSL.
Xiaochaihutangforliverdiseases:Aliteraturereview.
WorldJTraditChinMed2017;3:46-50.
1ThisisanopenaccessarticledistributedunderthetermsoftheCreativeCommonsAttributionNonCommercialShareAlike3.
0License,whichallowsotherstoremix,tweak,andbuildupontheworknoncommercially,aslongastheauthoriscreditedandthenewcreationsarelicensedundertheidenticalterms.
Forreprintscontact:reprints@medknow.
com2017WorldJournalofTraditionalChineseMedicine|PublishedbyWoltersKluwerMedknowReceived:01092016,Accepted:03052017WorldJTraditChinMed|Volume3|Issue3|JulySeptember201747patientswithchronichepatitisC,Yamashikietal.
[4]pointedoutthatXCHTcouldmediatethelevelsofIL4,IL5,andIL10topreventthediseaseprogression.
Inimprintingcontrolregion(ICR)micewithliverinjuryinducedbyDgalactosamine,oraladministrationofXCHTfor14daysresultedinasignificantdecreaseofIL6andTNFαlevelsintheserum,asimilarreductionofFasmRNA,FasLmRNA,andBaxproteinexpression,butanincreaseoftheBcl2mRNAexpressioninthelivertissues,andfinally,XCHTrelievedthiskindofliverinjurythroughthemechanismabove.
[5]HowXCHTanditscomponentsworkonthecountandproliferationofTcellsubsetsinspleniccellsandhepatocytesinvitrohasalsobeenexamined.
ThedataindicatedthatXCHTselectivelyinhibitedtheproliferationofCD8+TcelltoregulatetheCD4/CD8ratioduetotheconstituentwogonin7Oglucuronideanditsmetabolin,whichmighttakeanessentialroleinthetreatmentofchronichepatitis.
[6]Inotherwords,thetherapeuticeffectofXCHTmightbeimprovingbodyresistance,removingevilpathogen,andstrengtheningorregulatingimmunefunction.
[7]ThepotentsynergisticeffectofXCHTonopsonicintracavitaryofHBVcarrierhasalsobeenconfirmedinamousemodel,whichinspiresawidespreaduseofXCHTinimmunetherapies.
[8]Changetal.
[9]testedtheantiHBVactivityofXCHTinHepG(2)2.
2.
15cellmodelandfoundXCHTcouldbesupplementarytonucleotideanalogstominimizetherecurrenceofviremiaafteritsdiscontinuation;however,thiseffectmightnotbemediatedbysaikosaponinA.
Nonalcoholicfattyliverdisease(NAFLD)referstotheaccumulationofexcessivefatintheliverofapatientwhohasnohistoryofalcoholabuse.
Db/dbmicefedonamethionineandcholinedeficientdiet,asanimalmodelsofnonalcoholicsteatohepatitis(NASH),andweretreatedwithXCHTfor4weeks.
Afterbiochemical,pathological,andmolecularanalyses,itwasrevealedthatthevalueofserumalanineaminotransferase(ALT)andthedegreeoflivernecroinflammationandfibrosis,weredramaticallyimprovedbyXCHTandmeanwhilemalondialdehydelevelsinlivertissueswereloweraftertreatment.
AllthesesuggestedXCHTmightinhibithepaticnecroinflammationandfibrosisinpatientswithNASH.
[10]StudiesofNammi'slaboratoryreportedtheunderlyingmechanismsofgingerasonecomponentofXCHTformulainregulatinghepaticcholesterolandlipidmetabolismofhighfatdiet(HFD)fedrats,[11,12]whosehypercholesterolemiaweremainlyregulatedbyincreasedhepaticlowdensitylipoproteinreceptorandreduced3hydroxy3methylglutarylCoAreductase.
HFDinducedhepaticinflammationwasprovedtoattenuatebecauseofgingerextractthroughinhibitionofnuclearfactorkB(NFkB).
[13]Gaoetal.
verifiedthatethanolicextractofginger(50mg/kg)significantlyreduceddyslipidemiaandhepaticlipidaccumulationinfructoseinducedNASHbymodulationofthehepaticcarbohydrateresponseelementbindingproteinmediatedpathway.
[14]AntifibrogeniceffectsInratswhosebileductswereligated,XCHTwasfoundtoreducecholestasissignificantly,cutdownthecollagencontentby50%,andexertantifibrogeniceffectbydownregulatinghepaticmRNAexpressionofprocollagenalpha1typeI,typeIII,andtissueinhibitorsofmetalloproteinase(TIMP)1.
[15]Thebalanceofmatrixmetalloproteinases(MMPs)andTIMPswasthefurtherbreakthroughpointofresearchonXCHT.
MMPhaddegradativeactivityagainstcollagen,whereasTIMPcontrolledtheactiveformsofMMPbyblockingtheactivesiteofit.
Sakaidaetal.
[16]claimedXCHTraisedMMP2,13activitiesandinhibitedTIMP1,2activitiesofhepaticstellatecells(HSCs)probablybyP38pathway.
Inaninvitrostudy,[17]XCHTsignificantlyaccumulatedthestellatecells,transformingmorphologicallytomyofibroblastlikecellsintheG0/G1phase,anddecreasedcellnumbersubsequentlyinG2/Mphase.
FromfurtherinsightintoXCHT,baicalinandbaicaleinbelongingtoflavonoidshadtheabilitytoinhibittheactivationofHSCsagainstfibrosis.
[18,19]XCHTalsosuppressedfibrogenesisbyreductionoflipidperoxides,andthispreventiveeffectwasshownininhibitingbothlipidperoxidationinculturedrathepaticcellswhichwereinthestateofoxidativestress,andgenerationofalphasmoothmuscleactin,typeIcollagenexpression,cellmultiplication,andoxidativeburstinculturedratHSCsconsideredasthemaincollagenproducingcells.
[19,20]Inaddition,XCHTshoweditsdosedependentsuppressiontoFe2+/adenosine5'diphosphateinducedlipidperoxidationinrathepaticmitochondriaanditsabilityoffreeradicalelimination.
[19]Onoetal.
[21]holdedthatadministrationofXCHTbroughtaboutanincreaseinretinoidlevelandadecreaseinhydroxyprolinelevelofliver,inhibitingactivationofItocells,andthen,causinginhibitionofcollagenproductionandpreventionofliverfibrosis.
However,whenactiveconstituentsofXCHTweretakenalone,liverretinoidlevelsremainedlow,implyingthatitwasinteractionamongactiveconstituentsofXCHTthatsuppressedactivationofItocell.
[22]Table1:ComponentsandconstituentsofXiaoChaiHuTangComponentsConstituentsBupleurumrootSaikosides,quercetin,αchondrillasterol,polysaccharide,naphthasPinelliaternataβsitosterol,glucosides,aminoacids,proteins,naphthas,saponin,alkaloidsScutellariarootBaicalins,chrysin,wogonoside,neobaicalein,naphthas,aminoacidsJujubaefructusBetulinicacid,oleanolicacid,crategolicacid,triterpenoid,alkaloids,flavonoids,saccharides,vitaminsPanaxginsengGinsenosides,polysaccharide,αpanacene,panaxacid,panasenosideLicoriceGlycyrrhizinGingerNaphthas(includinggingerolandzingiberene),gingerolWorldJTraditChinMed|Volume3|Issue3|JulySeptember201748AntihepatomaeffectsXCHTinducedTNFαandGCSFinvitroinperipheralbloodmononuclearcellsofpatientswithhepatocellularcarcinoma(HCC),whichmightbethesourceofbenefitsforpatientstakingadministrationofXCHTtodealwithhepatoma.
[23]AsubsequentstudysuggestedthatXCHTpreventedhepatocarcinogenesiswithinhibitionof8hydroxy2'-deoxyguanosine(8OHdG)formation,whichplayedanimportantroleinoxidativestress.
[24]Meanwhile,theviciouscirclebetweentheoxidativestressandthealkalinephosphataseinactivationthroughlipopolysaccharides(LPS)catecholaminesinteractionsinthegut,liver,andbraincouldbeattenuatedbyXCHTduringCCI4+ethanolinducedmouseHCC.
[25]Besides,XCHTwasprovedtosuppresstheproliferationofthecarcinomacelllinesinmorphological,DNA,andcellcycleanalyses,inducingapoptosisintheearlyperiodofexposureandarrestattheG0/G1phaseinthelateperiodofexposure.
ThesuppressiveeffectofXCHTwasstrongerthaneachofitsmajoringredients,andalmostnoinhibitionwasobservedinnormalhumanperipheralbloodlymphocytesornormalrathepaticcells.
[26]Otherresearchesdemonstratedthatbaicalein,baicalin,andsaikosaponinAsuppressedculturedhumanhepatomacellproliferationinadosedependentmannerbutirrelevanttothecellcycle.
Furthermore,itwasreportedthatsaikosaponinApossessedstrongcellkillingability,butsaikosaponinC,ginsenosideRb1,andginsenosideRg1didnotworkoncellproliferation.
[27]Afterward,XCHTwasshowntoimprovetheimmunefunctionoftumorbearingmicetoinhibitthegrowthofsolidlivercancer.
[28]However,Watanabeetal.
[29]foundthenumberofGlutathioneStransferaseplacentalformpositiveareaswithintheHCClesionswassmallerinboththecontrolandthelycopenegroupthanthatintheXCHTgroup(P=0.
024andP=0.
012,respectively),whichmeantlongtermadministrationofXCHTdidnotreducetheriskofhepatocarcinogenesisinLong–Evanscinnamonrats.
HumanClinicalTrialsHepatitisInaninvivostudy,[30]XCHTseemedtohelpeliminateHBeAginchildren.
Amongthe24chronichepatitisCpatientswhowerenotcandidatesforIFNbasedtherapyandreceivedXCHTatthedoseof2.
5gthreetimesdailyfor12months,aspartateaminotransferaseof67%ofparticipants,ALTof75%ofparticipants,viralloadresponseof29%ofparticipantsandhistologyactivityindexscoresof38%ofparticipantswereimprovedaccordingtotheassay.
ThedatasupportedthenotionthatXCHTcouldimproveliverpathologyinselectedhepatitisCpatientswhowerenotcandidatesforIFNbasedtreatment.
[31]Anothertrial,inwhich26healthyparticipantsreceivedXCHTatthedoseof2.
5gtwicedailyfor5daysandunderwentthecaffeinetestonday1andday5,[32]showedthatthemeanactivityofcytochromeP450enzymes1A2(CYP1A2)reducedby16%onboth1stdayand5thdaycomparedwiththebaseline;themeanactivityofxanthineoxidase(XO)alsodramaticallyreducedby25%on1stdayand20%on5thdaycomparedwiththebaseline;theactivityofcytochromeP450enzymes3Atendedtobeloweron5thdaythanthebaseline.
Inbrief,XCHTreducedCYP1A2andXOactivityinhuman.
HepaticfibrosisTheherbalmedicineXCHThasbeenadministeredtoahugenumberofpatientswithchronicliverdiseases.
InpatientswithHCVpositivelivercirrhosis,thelifeextendingeffectofXCHTwasattributedtotwoofitssevenherbcomponents,thatwas,scutellariarootandlicorice.
ThemechanismmightincludethepromotionofIL12production.
[33]HepatomaXCHTisknowntodramaticallyinhibitdevelopmentofhepatomaandplaysaroleinlifeprolongation.
Okaetal.
[34]performedaprospective,randomized,nonblindcontrolledstudythatrevealedXCHTcouldhelptopreventtheprogressionofHCCinpatientswithcirrhosis,especiallyinpatientswithoutHBsantigen.
PotentialDrugInteractionBecauseofcurrentpopularityofXCHTinhepaticdisease,cancer,andotherdiseases,itisessentialtodeterminewhetherithasaneffectoncytochromeP450,thehepaticenzymesysteminchargeofmetabolismofmanymedications.
Nishimuraetal.
[35]tooktheattitudethatXCHTcutdownthebioavailabilityoftolbutamide(asulfonylureahypoglycemicagent)inratsafteroraladministration,butitmadenoeffectontheintravenousadministrationoftolbutamide.
Thatwastosay,theabovechangewasnotrelatedtohepaticmetabolism.
Simultaneously,theyclaimedthatXCHThadaninhibitiononthegastricemptyingandanincreaseontheintragastricpH,leadingtothelowerplasmaconcentrationoftolbutamideafteroraladministration,butitaffectedneithertheintragastricdissolutionnorthegastricabsorptionoftolbutamide.
ThisviewcontradictsthepreviousconclusionthatXCHTslightlyhastenedthegastrointestinalabsorptionoftolbutamide,whichmightenhancethehypoglycemiceffectofthesulfonylureaintheearlyperiodafteroraladministration.
[36,37]SideEffectsSomecasereportshaveshownthesideeffectsofXCHT,mostofwhichareinterstitialpneumoniaandacuterespiratoryfailureinJapan.
[38]Patientsoftenmanifestedwithcoughing,dyspnea,andacuteepisodesoffever.
Chestradiographsrevealeddiffusefrostedglassshadowsandinfiltration.
ThereweresignificantincreasesabovenormalinbothserumCreactiveproteinandlactatedehydrogenaselevel.
Hypoxiawascommon.
AnalysisofbronchoalveolarlavagefluidshowedabnormallyhighWorldJTraditChinMed|Volume3|Issue3|JulySeptember201749proportionsoflymphocytesandneutrophilsandalowCD4/CD8ratio.
[39]Asreportwent,theyoungestsufferingfrominterstitialpneumoniaattributedtoXCHTinJapanwasa7yearoldboywithacutelymphoblasticleukemiacomplicatedbytypeChepatitis.
[40]Atthesametime,druginducedpneumoniaduetoXCHToccurredina71yearoldwomanwithautoimmunehepatitis.
[41]Inanotherautopsycase,[42]itwasinferredthatthedevelopmentofinterstitialpneumoniawascausedbyHCVincombinationwithXCHTinducedlunginjury.
TherewasalsosomeonegettinghepaticinjuryandpneumonitissimultaneouslyafterintakeofXCHT.
[43]Asthesecasereportsshow,themorbidityandriskofXCHTinducedinterstitialpneumoniaareincreasedbycoadministrationofIFN,[44,45]durationofmedication,andhighinanelderlypopulation.
[38]Murakamietal.
[46]madeitclearthatinterstitialpneumoniawasasideeffectoftreatmentwithIFN,andXCHTmightpotentiatethissideeffect.
ApossiblemechanismwasthatXCHTcouldoverstimulatetheneutrophilswhichwascausedbyIFNtoaccumulateinthelung.
Activatedneutrophilsreleasedgranulocyteselastaseandoxygenradicalsthatmightdamagelungtissue.
Whenthefibroblastsrepairedthedamagedtissue,pulmonaryfibrosismightoccur.
Therefore,itissaferforXCHTtobeusedinpatientswithhepaticdiseasewhocontraindicateIFN.
AccordingtoSato,[39]themeantreatmentcourseofXCHTbeforetheoccurrenceofpneumonitiswas50.
2±42.
1daysandthemeanagewas63.
7yearold.
Nevertheless,Ohtakeetal.
[47]arguedthatglycyrrhizin,ametaboliteoflicoriceasoneoftheprimarycomponentsinXCHT,raisedinvitroIL6inantiCD3monoclonalantibody(antiCD3mAb)stimulatedlungmononuclearcellsinacelltypespecificanddosedependentmanner,soXCHTalleviatedLPSinducedlunginjuryatthelaterphasewhenlungleakwasobvious,butwasineffectiveonearlyneutrophilsequestrationtothelunginBALB/cmice.
ConclusionsandOtherPotentialUsesXCHT,atraditionalcommonlyappliedChineseherbalmedicineformula,haslongbeenusedtopatientswithhepaticdiseasesinAsia.
[48]Infact,XCHTstillhasmanyothereffectsthathavenotbeenexerted.
Inliverdiseases,moreandmorehumanclinicaltrialsareneededtoobtainenoughproofsfortheclinicaluseofXCHTinmoreaspects,suchasNAFLDandautoimmuneliverdisease.
XCHTexhibitsaverylowtoxicityprofileandappearstobesafeforclinicaluseandconsumption.
AcknowledgmentWewouldliketothankallthosewhohavecontributedtheirtimeandknowledgetothismanuscript.
WealsowishtothanktwoanonymousrefereesandtheacademiceditorsofWJTCMfortheirveryconstructivefeedback.
FinancialsupportandsponsorshipThisworkwassupportedbygrantsfromtheScienceandTechnologyCommissionFoundationofShanghaiMunicipality;China(grants13401902801).
ConflictsofinterestTherearenoconflictsofinterest.
References1.
YamashikiM,KosakaY,NishimuraA,TakaseK,IchidaF.
Efficacyofaherbalmedicine"Shosaikoto"ontheimprovementofimpairedcytokineproductionofperipheralbloodmononuclearcellsinpatientswithchronicviralhepatitis.
JClinLabImmunol1992;37:11121.
2.
YamashikiM,NishimuraA,SakaguchiS,SuzukiH,KosakaY.
EffectsoftheJapaneseherbalmedicine'Shosaikoto'asacytokineinducer.
EnvironToxicolPharmacol1996;2:3016.
3.
KakumuS,YoshiokaK,WakitaT,IshikawaT.
EffectsofTJ9Shosaikoto(kampomedicine)oninterferongammaandantibodyproductionspecificforhepatitisBvirusantigeninpatientswithtypeBchronichepatitis.
IntJImmunopharmacol1991;13:1416.
4.
YamashikiM,NishimuraA,SuzukiH,SakaguchiS,KosakaY.
EffectsoftheJapaneseherbalmedicine"Shosaikoto"(TJ9)oninvitrointerleukin10productionbyperipheralbloodmononuclearcellsofpatientswithchronichepatitisC.
Hepatology1997;25:13907.
5.
ZhouYX,QiuYQ,XuLQ,GuoJ,LiLJ.
XiaoChaiHuTangintreatingmodelmicewithDgalactosamineinducedliverinjury.
AfrJTraditComplementAlternMed2012;9:40511.
6.
OhtakeN,YamamotoM,TakedaS,AburadaM,IshigeA,WatanabeK,etal.
TheherbalmedicineShosaikotoselectivelyinhibitsCD8+Tcellproliferation.
EurJPharmacol2005;507:30110.
7.
LiuZ,XiongM,ZhangH.
ExperimentalstudyoninhibitoryeffectofxiaochaihudecoctiononduckhepatitisBvirus.
ZhongguoZhongXiYiJieHeZaZhi2000;20:8535.
8.
AkbarSM,YamamotoK,AbeM,NinomiyaT,TanimotoK,MasumotoT,etal.
PotentsynergisticeffectofShosaikoto,aherbalmedicine,duringvaccinetherapyinamurinemodelofhepatitisBviruscarrier.
EurJClinInvest1999;29:78692.
9.
ChangJS,WangKC,LiuHW,ChenMC,ChiangLC,LinCC.
Shosaikoto(XiaoChaiHuTang)andcrudesaikosaponinsinhibithepatitisBvirusinastableHBVproducingcellline.
AmJChinMed2007;35:34151.
10.
TakahashiY,SoejimaY,KumagaiA,WatanabeM,UozakiH,FukusatoT.
InhibitoryeffectsofJapaneseherbalmedicinesShosaikotoandjuzentaihotoonnonalcoholicsteatohepatitisinmice.
PLoSOne2014;9:e87279.
11.
NammiS,SreemantulaS,RoufogalisBD.
ProtectiveeffectsofethanolicextractofZingiberofficinalerhizomeonthedevelopmentofmetabolicsyndromeinhighfatdietfedrats.
BasicClinPharmacolToxicol2009;104:36673.
12.
NammiS,KimMS,GavandeNS,LiGQ,RoufogalisBD.
Regulationoflowdensitylipoproteinreceptorand3hydroxy3methylglutarylcoenzymeAreductaseexpressionbyZingiberofficinaleintheliverofhighfatdietfedrats.
BasicClinPharmacolToxicol2010;106:38995.
13.
LiXH,McGrathKC,NammiS,HeatherAK,RoufogalisBD.
AttenuationofliverproinflammatoryresponsesbyZingiberofficinaleviainhibitionofNFkappaBactivationinhighfatdietfedrats.
BasicClinPharmacolToxicol2012;110:23844.
14.
GaoH,GuanT,LiC,ZuoG,YamaharaJ,WangJ,etal.
TreatmentwithgingeramelioratesfructoseinducedFattyliverandhypertriglyceridemiainrats:Modulationofthehepaticcarbohydrateresponseelementbindingproteinmediatedpathway.
EvidBasedComplementAlternatMed2012;2012:570948.
15.
ChenMH,ChenJC,TsaiCC,WangWC,ChangDC,LinCC,etal.
Shosaikotopreventsliverfibrosisinducedbybileductligationinrats.
AmJChinMed2004;32:195207.
16.
SakaidaI,HironakaK,KimuraT,TeraiS,YamasakiT,OkitaK.
HerbalmedicineShosaikoto(TJ9)increasesexpressionmatrixmetalloproteinases(MMPs)withreducedexpressionoftissueinhibitorofmetalloproteinases(TIMPs)inratstellatecell.
LifeSci2004;74:225163.
17.
KayanoK,SakaidaI,UchidaK,OkitaK.
InhibitoryeffectsoftheherbalmedicineShosaikoto(TJ9)oncellproliferationandprocollagenWorldJTraditChinMed|Volume3|Issue3|JulySeptember201750geneexpressionsinculturedrathepaticstellatecells.
JHepatol1998;29:6429.
18.
ShimizuI.
Shosaikoto:Japaneseherbalmedicineforprotectionagainsthepaticfibrosisandcarcinoma.
JGastroenterolHepatol2000;15Suppl:D8490.
19.
ShimizuI,MaYR,MizobuchiY,LiuF,MiuraT,NakaiY,etal.
EffectsofShosaikoto,aJapaneseherbalmedicine,onhepaticfibrosisinrats.
Hepatology1999;29:14960.
20.
KitadeY,WatanabeS,MasakiT,NishiokaM,NishinoH.
InhibitionofliverfibrosisinLECratsbyacarotenoid,lycopene,oraherbalmedicine,Shosaikoto.
HepatolRes2002;22:196205.
21.
OnoM,MiyamuraM,KyotaniS,SaibaraT,OhnishiS,NishiokaY.
EffectsofShosaikotoextractonliverfibrosisinrelationtothechangesinhydroxyprolineandretinoidlevelsoftheliverinrats.
JPharmPharmacol1999;51:107984.
22.
MiyamuraM,OnoM,KyotaniS,NishiokaY.
EffectsofShosaikotoextractonfibrosisandregenerationoftheliverinrats.
JPharmPharmacol1998;50:97105.
23.
YamashikiM,NishimuraA,NomotoM,SuzukiH,KosakaY.
Herbalmedicine'Shosaikoto'inducestumournecrosisfactoralphaandgranulocytecolonystimulatingfactorinvitroinperipheralbloodmononuclearcellsofpatientswithhepatocellularcarcinoma.
JGastroenterolHepatol1996;11:13742.
24.
ShiotaG,MaetaY,MukoyamaT,YanagidaniA,UdagawaA,OyamaK,etal.
EffectsofShoSaikotoonhepatocarcinogenesisand8hydroxy2'deoxyguanosineformation.
Hepatology2002;35:112533.
25.
LiuXQ,HuXJ,XuHX,ZengXY.
XiaochaihuDecoctionattenuatestheviciouscirclebetweentheoxidativestressandtheALPinactivationthroughLPScatecholaminesinteractionsingut,liverandbrainduringCCI4ethanolinducedmouseHCC.
BMCComplementAlternMed2013;13:375.
26.
YanoH,MizoguchiA,FukudaK,HaramakiM,OgasawaraS,MomosakiS,etal.
TheherbalmedicineshosaikotoinhibitsproliferationofcancercelllinesbyinducingapoptosisandarrestattheG0/G1phase.
CancerRes1994;54:44854.
27.
OkitaK,LiQ,MurakamioT,TakahashiM.
AntigrowtheffectswithcomponentsofShosaikoto(TJ9)onculturedhumanhepatomacells.
EurJCancerPrev1993;2:16975.
28.
LiJ,XieM,GanY.
EffectofXiaochaihudecoctionanddifferentherbalformulationofcomponentoninhibitingH22livercancerinmiceandenhancingimmunefunction.
ZhongguoZhongYaoZaZhi2008;33:103944.
29.
WatanabeS,KitadeY,MasakiT,NishiokaM,SatohK,NishinoH.
EffectsoflycopeneandShosaikotoonhepatocarcinogenesisinaratmodelofspontaneouslivercancer.
NutrCancer2001;39:96101.
30.
TajiriH,KozaiwaK,OzakiY,MikiK,ShimuzuK,OkadaS.
Effectofshosaikoto(xiaochaihutang)onHBeAgclearanceinchildrenwithchronichepatitisBvirusinfectionandwithsustainedliverdisease.
AmJChinMed1991;19:1219.
31.
DengG,KurtzRC,VickersA,LauN,YeungKS,ShiaJ,etal.
AsinglearmphaseIIstudyofaFarEasterntraditionalherbalformulation(Shosaikotoorxiaochaihutang)inchronichepatitisCpatients.
JEthnopharmacol2011;136:837.
32.
SaruwatariJ,NakagawaK,ShindoJ,NachiS,EchizenH,IshizakiT.
TheinvivoeffectsofShosaikoto,atraditionalChineseherbalmedicine,ontwocytochromeP450enzymes(1A2and3A)andxanthineoxidaseinman.
JPharmPharmacol2003;55:15539.
33.
YamashikiM,NishimuraA,HuangXX,NoboriT,SakaguchiS,SuzukiH.
EffectsoftheJapaneseherbalmedicine"Shosaikoto"(TJ9)oninterleukin12productioninpatientswithHCVpositivelivercirrhosis.
DevImmunol1999;7:1722.
34.
OkaH,YamamotoS,KurokiT,HariharaS,MarumoT,KimSR,etal.
ProspectivestudyofchemopreventionofhepatocellularcarcinomawithShosaikoto(TJ9).
Cancer1995;76:7439.
35.
NishimuraN,NaoraK,HiranoH,IwamotoK.
AChinesetraditionalmedicine,shosaikoto(xiaochaihutang),reducesthebioavailabilityoftolbutamideafteroraladministrationinrats.
AmJChinMed1999;27:35563.
36.
NishimuraN,NaoraK,HiranoH,IwamotoK.
Effectsofshosaikoto(xiaochaihutang),aChinesetraditionalmedicine,onthegastricfunctionandabsorptionoftolbutamideinrats.
YakugakuZasshi2001;121:1539.
37.
NishimuraN,NaoraK,HiranoH,IwamotoK.
EffectsofShosaikotoonthepharmacokineticsandpharmacodynamicsoftolbutamideinrats.
JPharmPharmacol1998;50:2316.
38.
LeeJK,KimJH,ShinHK.
TherapeuticeffectsoftheorientalherbalmedicineShosaikotoonlivercirrhosisandcarcinoma.
HepatolRes2011;41:82537.
39.
SatoA,ToyoshimaM,KondoA,OhtaK,SatoH,OhsumiA.
PneumonitisinducedbytheherbalmedicineShosaikotoinJapan.
NihonKyobuShikkanGakkaiZasshi1997;35:3915.
40.
WadaY,KuboM.
AcutelymphoblasticleukemiacomplicatedbytypeChepatitisduringtreatmentandfurtherbyacuteinterstitialpneumoniaduetoShosaikotoin7yearold.
Arerugi1997;46:114855.
41.
KatouK,MoriK.
AutoimmunehepatitiswithdruginducedpneumoniaduetoShosaikoto.
NihonKokyukiGakkaiZasshi1999;37:6416.
42.
TomiokaH,HashimotoK,OhnishiH,FujiyamaR,SakuraiT,TadaK,etal.
AnautopsycaseofinterstitialpneumoniaprobablyinducedbyShosaikoto.
NihonKokyukiGakkaiZasshi1999;37:10138.
43.
DaiboA,YoshidaY,KitazawaS,KosakaY,BandoT,SudoM.
Acaseofpneumonitisandhepaticinjurycausedbyaherbaldrug(shosaikoto).
NihonKyobuShikkanGakkaiZasshi1992;30:15838.
44.
TojimaH,YamazakiT,TokudomeT.
TwocasesofpneumoniacausedbyShosaikoto.
NihonKyobuShikkanGakkaiZasshi1996;34:90410.
45.
NakagawaA,YamaguchiT,TakaoT,AmanoH.
FivecasesofdruginducedpneumonitisduetoShosaikotoorinterferonalphaorboth.
NihonKyobuShikkanGakkaiZasshi1995;33:13616.
46.
MurakamiK,OkajimaK,SakataK,TakatsukiK.
Apossiblemechanismofinterstitialpneumoniaduringinterferontherapywithshosaikoto.
NihonKyobuShikkanGakkaiZasshi1995;33:38994.
47.
OhtakeN,SuzukiR,DaikuharaH,NakaiY,YamamotoM,AmagayaS,etal.
ModulationoflunglocalimmuneresponsesbyoraladministrationofaherbalmedicineShosaikoto.
IntJImmunopharmacol2000;22:41930.
48.
ChenYW,TsaiMY,PanHB,TsengHH,HungYT,ChouCP.
GadoxeticacidenhancedMRIandsonoelastography:NoninvasiveassessmentsofchemopreventionofliverfibrosisinthioacetamideinducedratswithShoSaikoTo.
PLoSOne2014;9:e114756.
Table1liststheprincipalconstituentsofeachherbcomponent.
ThisformulaisderivedfromthebookTreatiseonColdDamageDiseases,writtenbyZhangZhongjing(afamousphysicianofthelateEasternHanDynasty,about200A.
D.
),andnow,manyexperimentalandclinicalresearchesincludingprospective,randomized,andplacebocontrolledtrialshaveinvolvedtheclinicefficacyofXCHT.
Althoughithasbeenwidelyusedbypatientswithdifferentkindsofhepaticdiseases,suchashepatitisandliverfibrosis,itsbenefitsclaimedneedanaccurateassessment.
ThisreviewsumsuptheavailablescientificfindingsaboutXCHTandhepaticdiseases,pointingoutthedirectionforfurtherstudies.
MechanismsofActionHepatoprotectiveeffectsXCHThelpstomaintainthebalanceamongtheimmunologicalcytokines,whichmayhaveimmunologicalbenefitstochronicviralhepatitis.
[1]Forexample,scutellariarootandlicoriceinXCHTcouldinducetheproductionofcytokinessuchasinterleukin(IL)1β,tumornecrosisfactor(TNF)α,andgranulocytecolonystimulatingfactor(GCSF)frommonocytes/macrophages.
[2]Kakumuetal.
[3]foundXCHTincreasedinterferonγ(IFNγ)andantibodyagainstHBV,strengtheningcellularandhumoralimmuneresponses.
ToXiaoChaiHuTangforLiverDiseases:ALiteratureReviewYiWanga,LiLib,YanMeiChenga,ShengLiangZhuaaDepartmentofGastroenterology,YueyangHospitalofIntegratedTraditionalChineseandWesternMedicine,ShanghaiUniversityofTraditionalChineseMedicine,bClinicalResearchInstituteofIntegratedTraditionalChineseandWesternMedicine,YueyangHospitalofIntegratedTraditionalChineseandWesternMedicine,ShanghaiUniversityofTraditionalChineseMedicine,Shanghai200437,ChinaTheobjectiveofthisstudyistosummarizethepharmacologicaleffectsandthemechanismsofactionofXiaoChaiHuTang(XCHT,MinorBupleurumDecoction)onliverdiseases,soastogiverelevantresearchersavaluableinsightandbenefitpatientswithhepatopathy.
PubMedwasusedtosearchforandcollectscientificpublicationsrelatedtoXCHTandliverdiseasesfrom1986to2016.
Theavailablescientificresultsorevidencewereread,classified,andanalyzed.
XCHTshowedclinicalefficacyinpatientswithhepaticdiseasesincludinghepatitis,hepaticfibrosis,andhepatoma.
Themechanismsinvolvedtheproductionofcytokines,theregulationofimmunefunction,thesuppressionoflipidperoxidation,etc.
,XCHTmightworkonthemetabolismofsomemedicationssuchastolbutamidebytheregulationofgastricemptyingandintragastricpH.
XCHTexhibitedaverylowtoxicityprofile,suchasinterstitialpneumoniaduetodurationofmedication,patients'age,anddrugcombination.
XCHThasbeenaeutherapeuticsupplementalremedyforliverdiseases.
However,manymechanismsofactionandeffectsofXCHTonnewtypesofliverdiseasesstillremainunclear,somoreandmoreanimalexperimentsandhumanclinicaltrialsareneededtoobtainenoughproofsfortheclinicaluseofXCHTinnewtypesofhepatosissuchasnonalcoholicfattyliverdiseaseandautoimmuneliverdisease.
Keywords:Hepaticfibrosis,hepatitis,hepatoma,interstitialpneumonia,tolbutamide,XiaoChaiHuTang(minorbupleurumdecoction)AccessthisarticleonlineQuickResponseCode:Website:www.
wjtcm.
netDOI:10.
4103/wjtcm.
wjtcm_4_17AbstractDownloadfreefromwww.
wjtcm.
netAddressforcorrespondence:Prof.
ShengLiangZhu,DepartmentofGastroenterology,YueyangHospitalofIntegratedTraditionalChineseandWesternMedicine,ShanghaiUniversityofTraditionalChineseMedicine,Shanghai200437,China.
Email:zhushengliang999@126.
comHowtocitethisarticle:WangY,LiL,ChengYM,ZhuSL.
Xiaochaihutangforliverdiseases:Aliteraturereview.
WorldJTraditChinMed2017;3:46-50.
1ThisisanopenaccessarticledistributedunderthetermsoftheCreativeCommonsAttributionNonCommercialShareAlike3.
0License,whichallowsotherstoremix,tweak,andbuildupontheworknoncommercially,aslongastheauthoriscreditedandthenewcreationsarelicensedundertheidenticalterms.
Forreprintscontact:reprints@medknow.
com2017WorldJournalofTraditionalChineseMedicine|PublishedbyWoltersKluwerMedknowReceived:01092016,Accepted:03052017WorldJTraditChinMed|Volume3|Issue3|JulySeptember201747patientswithchronichepatitisC,Yamashikietal.
[4]pointedoutthatXCHTcouldmediatethelevelsofIL4,IL5,andIL10topreventthediseaseprogression.
Inimprintingcontrolregion(ICR)micewithliverinjuryinducedbyDgalactosamine,oraladministrationofXCHTfor14daysresultedinasignificantdecreaseofIL6andTNFαlevelsintheserum,asimilarreductionofFasmRNA,FasLmRNA,andBaxproteinexpression,butanincreaseoftheBcl2mRNAexpressioninthelivertissues,andfinally,XCHTrelievedthiskindofliverinjurythroughthemechanismabove.
[5]HowXCHTanditscomponentsworkonthecountandproliferationofTcellsubsetsinspleniccellsandhepatocytesinvitrohasalsobeenexamined.
ThedataindicatedthatXCHTselectivelyinhibitedtheproliferationofCD8+TcelltoregulatetheCD4/CD8ratioduetotheconstituentwogonin7Oglucuronideanditsmetabolin,whichmighttakeanessentialroleinthetreatmentofchronichepatitis.
[6]Inotherwords,thetherapeuticeffectofXCHTmightbeimprovingbodyresistance,removingevilpathogen,andstrengtheningorregulatingimmunefunction.
[7]ThepotentsynergisticeffectofXCHTonopsonicintracavitaryofHBVcarrierhasalsobeenconfirmedinamousemodel,whichinspiresawidespreaduseofXCHTinimmunetherapies.
[8]Changetal.
[9]testedtheantiHBVactivityofXCHTinHepG(2)2.
2.
15cellmodelandfoundXCHTcouldbesupplementarytonucleotideanalogstominimizetherecurrenceofviremiaafteritsdiscontinuation;however,thiseffectmightnotbemediatedbysaikosaponinA.
Nonalcoholicfattyliverdisease(NAFLD)referstotheaccumulationofexcessivefatintheliverofapatientwhohasnohistoryofalcoholabuse.
Db/dbmicefedonamethionineandcholinedeficientdiet,asanimalmodelsofnonalcoholicsteatohepatitis(NASH),andweretreatedwithXCHTfor4weeks.
Afterbiochemical,pathological,andmolecularanalyses,itwasrevealedthatthevalueofserumalanineaminotransferase(ALT)andthedegreeoflivernecroinflammationandfibrosis,weredramaticallyimprovedbyXCHTandmeanwhilemalondialdehydelevelsinlivertissueswereloweraftertreatment.
AllthesesuggestedXCHTmightinhibithepaticnecroinflammationandfibrosisinpatientswithNASH.
[10]StudiesofNammi'slaboratoryreportedtheunderlyingmechanismsofgingerasonecomponentofXCHTformulainregulatinghepaticcholesterolandlipidmetabolismofhighfatdiet(HFD)fedrats,[11,12]whosehypercholesterolemiaweremainlyregulatedbyincreasedhepaticlowdensitylipoproteinreceptorandreduced3hydroxy3methylglutarylCoAreductase.
HFDinducedhepaticinflammationwasprovedtoattenuatebecauseofgingerextractthroughinhibitionofnuclearfactorkB(NFkB).
[13]Gaoetal.
verifiedthatethanolicextractofginger(50mg/kg)significantlyreduceddyslipidemiaandhepaticlipidaccumulationinfructoseinducedNASHbymodulationofthehepaticcarbohydrateresponseelementbindingproteinmediatedpathway.
[14]AntifibrogeniceffectsInratswhosebileductswereligated,XCHTwasfoundtoreducecholestasissignificantly,cutdownthecollagencontentby50%,andexertantifibrogeniceffectbydownregulatinghepaticmRNAexpressionofprocollagenalpha1typeI,typeIII,andtissueinhibitorsofmetalloproteinase(TIMP)1.
[15]Thebalanceofmatrixmetalloproteinases(MMPs)andTIMPswasthefurtherbreakthroughpointofresearchonXCHT.
MMPhaddegradativeactivityagainstcollagen,whereasTIMPcontrolledtheactiveformsofMMPbyblockingtheactivesiteofit.
Sakaidaetal.
[16]claimedXCHTraisedMMP2,13activitiesandinhibitedTIMP1,2activitiesofhepaticstellatecells(HSCs)probablybyP38pathway.
Inaninvitrostudy,[17]XCHTsignificantlyaccumulatedthestellatecells,transformingmorphologicallytomyofibroblastlikecellsintheG0/G1phase,anddecreasedcellnumbersubsequentlyinG2/Mphase.
FromfurtherinsightintoXCHT,baicalinandbaicaleinbelongingtoflavonoidshadtheabilitytoinhibittheactivationofHSCsagainstfibrosis.
[18,19]XCHTalsosuppressedfibrogenesisbyreductionoflipidperoxides,andthispreventiveeffectwasshownininhibitingbothlipidperoxidationinculturedrathepaticcellswhichwereinthestateofoxidativestress,andgenerationofalphasmoothmuscleactin,typeIcollagenexpression,cellmultiplication,andoxidativeburstinculturedratHSCsconsideredasthemaincollagenproducingcells.
[19,20]Inaddition,XCHTshoweditsdosedependentsuppressiontoFe2+/adenosine5'diphosphateinducedlipidperoxidationinrathepaticmitochondriaanditsabilityoffreeradicalelimination.
[19]Onoetal.
[21]holdedthatadministrationofXCHTbroughtaboutanincreaseinretinoidlevelandadecreaseinhydroxyprolinelevelofliver,inhibitingactivationofItocells,andthen,causinginhibitionofcollagenproductionandpreventionofliverfibrosis.
However,whenactiveconstituentsofXCHTweretakenalone,liverretinoidlevelsremainedlow,implyingthatitwasinteractionamongactiveconstituentsofXCHTthatsuppressedactivationofItocell.
[22]Table1:ComponentsandconstituentsofXiaoChaiHuTangComponentsConstituentsBupleurumrootSaikosides,quercetin,αchondrillasterol,polysaccharide,naphthasPinelliaternataβsitosterol,glucosides,aminoacids,proteins,naphthas,saponin,alkaloidsScutellariarootBaicalins,chrysin,wogonoside,neobaicalein,naphthas,aminoacidsJujubaefructusBetulinicacid,oleanolicacid,crategolicacid,triterpenoid,alkaloids,flavonoids,saccharides,vitaminsPanaxginsengGinsenosides,polysaccharide,αpanacene,panaxacid,panasenosideLicoriceGlycyrrhizinGingerNaphthas(includinggingerolandzingiberene),gingerolWorldJTraditChinMed|Volume3|Issue3|JulySeptember201748AntihepatomaeffectsXCHTinducedTNFαandGCSFinvitroinperipheralbloodmononuclearcellsofpatientswithhepatocellularcarcinoma(HCC),whichmightbethesourceofbenefitsforpatientstakingadministrationofXCHTtodealwithhepatoma.
[23]AsubsequentstudysuggestedthatXCHTpreventedhepatocarcinogenesiswithinhibitionof8hydroxy2'-deoxyguanosine(8OHdG)formation,whichplayedanimportantroleinoxidativestress.
[24]Meanwhile,theviciouscirclebetweentheoxidativestressandthealkalinephosphataseinactivationthroughlipopolysaccharides(LPS)catecholaminesinteractionsinthegut,liver,andbraincouldbeattenuatedbyXCHTduringCCI4+ethanolinducedmouseHCC.
[25]Besides,XCHTwasprovedtosuppresstheproliferationofthecarcinomacelllinesinmorphological,DNA,andcellcycleanalyses,inducingapoptosisintheearlyperiodofexposureandarrestattheG0/G1phaseinthelateperiodofexposure.
ThesuppressiveeffectofXCHTwasstrongerthaneachofitsmajoringredients,andalmostnoinhibitionwasobservedinnormalhumanperipheralbloodlymphocytesornormalrathepaticcells.
[26]Otherresearchesdemonstratedthatbaicalein,baicalin,andsaikosaponinAsuppressedculturedhumanhepatomacellproliferationinadosedependentmannerbutirrelevanttothecellcycle.
Furthermore,itwasreportedthatsaikosaponinApossessedstrongcellkillingability,butsaikosaponinC,ginsenosideRb1,andginsenosideRg1didnotworkoncellproliferation.
[27]Afterward,XCHTwasshowntoimprovetheimmunefunctionoftumorbearingmicetoinhibitthegrowthofsolidlivercancer.
[28]However,Watanabeetal.
[29]foundthenumberofGlutathioneStransferaseplacentalformpositiveareaswithintheHCClesionswassmallerinboththecontrolandthelycopenegroupthanthatintheXCHTgroup(P=0.
024andP=0.
012,respectively),whichmeantlongtermadministrationofXCHTdidnotreducetheriskofhepatocarcinogenesisinLong–Evanscinnamonrats.
HumanClinicalTrialsHepatitisInaninvivostudy,[30]XCHTseemedtohelpeliminateHBeAginchildren.
Amongthe24chronichepatitisCpatientswhowerenotcandidatesforIFNbasedtherapyandreceivedXCHTatthedoseof2.
5gthreetimesdailyfor12months,aspartateaminotransferaseof67%ofparticipants,ALTof75%ofparticipants,viralloadresponseof29%ofparticipantsandhistologyactivityindexscoresof38%ofparticipantswereimprovedaccordingtotheassay.
ThedatasupportedthenotionthatXCHTcouldimproveliverpathologyinselectedhepatitisCpatientswhowerenotcandidatesforIFNbasedtreatment.
[31]Anothertrial,inwhich26healthyparticipantsreceivedXCHTatthedoseof2.
5gtwicedailyfor5daysandunderwentthecaffeinetestonday1andday5,[32]showedthatthemeanactivityofcytochromeP450enzymes1A2(CYP1A2)reducedby16%onboth1stdayand5thdaycomparedwiththebaseline;themeanactivityofxanthineoxidase(XO)alsodramaticallyreducedby25%on1stdayand20%on5thdaycomparedwiththebaseline;theactivityofcytochromeP450enzymes3Atendedtobeloweron5thdaythanthebaseline.
Inbrief,XCHTreducedCYP1A2andXOactivityinhuman.
HepaticfibrosisTheherbalmedicineXCHThasbeenadministeredtoahugenumberofpatientswithchronicliverdiseases.
InpatientswithHCVpositivelivercirrhosis,thelifeextendingeffectofXCHTwasattributedtotwoofitssevenherbcomponents,thatwas,scutellariarootandlicorice.
ThemechanismmightincludethepromotionofIL12production.
[33]HepatomaXCHTisknowntodramaticallyinhibitdevelopmentofhepatomaandplaysaroleinlifeprolongation.
Okaetal.
[34]performedaprospective,randomized,nonblindcontrolledstudythatrevealedXCHTcouldhelptopreventtheprogressionofHCCinpatientswithcirrhosis,especiallyinpatientswithoutHBsantigen.
PotentialDrugInteractionBecauseofcurrentpopularityofXCHTinhepaticdisease,cancer,andotherdiseases,itisessentialtodeterminewhetherithasaneffectoncytochromeP450,thehepaticenzymesysteminchargeofmetabolismofmanymedications.
Nishimuraetal.
[35]tooktheattitudethatXCHTcutdownthebioavailabilityoftolbutamide(asulfonylureahypoglycemicagent)inratsafteroraladministration,butitmadenoeffectontheintravenousadministrationoftolbutamide.
Thatwastosay,theabovechangewasnotrelatedtohepaticmetabolism.
Simultaneously,theyclaimedthatXCHThadaninhibitiononthegastricemptyingandanincreaseontheintragastricpH,leadingtothelowerplasmaconcentrationoftolbutamideafteroraladministration,butitaffectedneithertheintragastricdissolutionnorthegastricabsorptionoftolbutamide.
ThisviewcontradictsthepreviousconclusionthatXCHTslightlyhastenedthegastrointestinalabsorptionoftolbutamide,whichmightenhancethehypoglycemiceffectofthesulfonylureaintheearlyperiodafteroraladministration.
[36,37]SideEffectsSomecasereportshaveshownthesideeffectsofXCHT,mostofwhichareinterstitialpneumoniaandacuterespiratoryfailureinJapan.
[38]Patientsoftenmanifestedwithcoughing,dyspnea,andacuteepisodesoffever.
Chestradiographsrevealeddiffusefrostedglassshadowsandinfiltration.
ThereweresignificantincreasesabovenormalinbothserumCreactiveproteinandlactatedehydrogenaselevel.
Hypoxiawascommon.
AnalysisofbronchoalveolarlavagefluidshowedabnormallyhighWorldJTraditChinMed|Volume3|Issue3|JulySeptember201749proportionsoflymphocytesandneutrophilsandalowCD4/CD8ratio.
[39]Asreportwent,theyoungestsufferingfrominterstitialpneumoniaattributedtoXCHTinJapanwasa7yearoldboywithacutelymphoblasticleukemiacomplicatedbytypeChepatitis.
[40]Atthesametime,druginducedpneumoniaduetoXCHToccurredina71yearoldwomanwithautoimmunehepatitis.
[41]Inanotherautopsycase,[42]itwasinferredthatthedevelopmentofinterstitialpneumoniawascausedbyHCVincombinationwithXCHTinducedlunginjury.
TherewasalsosomeonegettinghepaticinjuryandpneumonitissimultaneouslyafterintakeofXCHT.
[43]Asthesecasereportsshow,themorbidityandriskofXCHTinducedinterstitialpneumoniaareincreasedbycoadministrationofIFN,[44,45]durationofmedication,andhighinanelderlypopulation.
[38]Murakamietal.
[46]madeitclearthatinterstitialpneumoniawasasideeffectoftreatmentwithIFN,andXCHTmightpotentiatethissideeffect.
ApossiblemechanismwasthatXCHTcouldoverstimulatetheneutrophilswhichwascausedbyIFNtoaccumulateinthelung.
Activatedneutrophilsreleasedgranulocyteselastaseandoxygenradicalsthatmightdamagelungtissue.
Whenthefibroblastsrepairedthedamagedtissue,pulmonaryfibrosismightoccur.
Therefore,itissaferforXCHTtobeusedinpatientswithhepaticdiseasewhocontraindicateIFN.
AccordingtoSato,[39]themeantreatmentcourseofXCHTbeforetheoccurrenceofpneumonitiswas50.
2±42.
1daysandthemeanagewas63.
7yearold.
Nevertheless,Ohtakeetal.
[47]arguedthatglycyrrhizin,ametaboliteoflicoriceasoneoftheprimarycomponentsinXCHT,raisedinvitroIL6inantiCD3monoclonalantibody(antiCD3mAb)stimulatedlungmononuclearcellsinacelltypespecificanddosedependentmanner,soXCHTalleviatedLPSinducedlunginjuryatthelaterphasewhenlungleakwasobvious,butwasineffectiveonearlyneutrophilsequestrationtothelunginBALB/cmice.
ConclusionsandOtherPotentialUsesXCHT,atraditionalcommonlyappliedChineseherbalmedicineformula,haslongbeenusedtopatientswithhepaticdiseasesinAsia.
[48]Infact,XCHTstillhasmanyothereffectsthathavenotbeenexerted.
Inliverdiseases,moreandmorehumanclinicaltrialsareneededtoobtainenoughproofsfortheclinicaluseofXCHTinmoreaspects,suchasNAFLDandautoimmuneliverdisease.
XCHTexhibitsaverylowtoxicityprofileandappearstobesafeforclinicaluseandconsumption.
AcknowledgmentWewouldliketothankallthosewhohavecontributedtheirtimeandknowledgetothismanuscript.
WealsowishtothanktwoanonymousrefereesandtheacademiceditorsofWJTCMfortheirveryconstructivefeedback.
FinancialsupportandsponsorshipThisworkwassupportedbygrantsfromtheScienceandTechnologyCommissionFoundationofShanghaiMunicipality;China(grants13401902801).
ConflictsofinterestTherearenoconflictsofinterest.
References1.
YamashikiM,KosakaY,NishimuraA,TakaseK,IchidaF.
Efficacyofaherbalmedicine"Shosaikoto"ontheimprovementofimpairedcytokineproductionofperipheralbloodmononuclearcellsinpatientswithchronicviralhepatitis.
JClinLabImmunol1992;37:11121.
2.
YamashikiM,NishimuraA,SakaguchiS,SuzukiH,KosakaY.
EffectsoftheJapaneseherbalmedicine'Shosaikoto'asacytokineinducer.
EnvironToxicolPharmacol1996;2:3016.
3.
KakumuS,YoshiokaK,WakitaT,IshikawaT.
EffectsofTJ9Shosaikoto(kampomedicine)oninterferongammaandantibodyproductionspecificforhepatitisBvirusantigeninpatientswithtypeBchronichepatitis.
IntJImmunopharmacol1991;13:1416.
4.
YamashikiM,NishimuraA,SuzukiH,SakaguchiS,KosakaY.
EffectsoftheJapaneseherbalmedicine"Shosaikoto"(TJ9)oninvitrointerleukin10productionbyperipheralbloodmononuclearcellsofpatientswithchronichepatitisC.
Hepatology1997;25:13907.
5.
ZhouYX,QiuYQ,XuLQ,GuoJ,LiLJ.
XiaoChaiHuTangintreatingmodelmicewithDgalactosamineinducedliverinjury.
AfrJTraditComplementAlternMed2012;9:40511.
6.
OhtakeN,YamamotoM,TakedaS,AburadaM,IshigeA,WatanabeK,etal.
TheherbalmedicineShosaikotoselectivelyinhibitsCD8+Tcellproliferation.
EurJPharmacol2005;507:30110.
7.
LiuZ,XiongM,ZhangH.
ExperimentalstudyoninhibitoryeffectofxiaochaihudecoctiononduckhepatitisBvirus.
ZhongguoZhongXiYiJieHeZaZhi2000;20:8535.
8.
AkbarSM,YamamotoK,AbeM,NinomiyaT,TanimotoK,MasumotoT,etal.
PotentsynergisticeffectofShosaikoto,aherbalmedicine,duringvaccinetherapyinamurinemodelofhepatitisBviruscarrier.
EurJClinInvest1999;29:78692.
9.
ChangJS,WangKC,LiuHW,ChenMC,ChiangLC,LinCC.
Shosaikoto(XiaoChaiHuTang)andcrudesaikosaponinsinhibithepatitisBvirusinastableHBVproducingcellline.
AmJChinMed2007;35:34151.
10.
TakahashiY,SoejimaY,KumagaiA,WatanabeM,UozakiH,FukusatoT.
InhibitoryeffectsofJapaneseherbalmedicinesShosaikotoandjuzentaihotoonnonalcoholicsteatohepatitisinmice.
PLoSOne2014;9:e87279.
11.
NammiS,SreemantulaS,RoufogalisBD.
ProtectiveeffectsofethanolicextractofZingiberofficinalerhizomeonthedevelopmentofmetabolicsyndromeinhighfatdietfedrats.
BasicClinPharmacolToxicol2009;104:36673.
12.
NammiS,KimMS,GavandeNS,LiGQ,RoufogalisBD.
Regulationoflowdensitylipoproteinreceptorand3hydroxy3methylglutarylcoenzymeAreductaseexpressionbyZingiberofficinaleintheliverofhighfatdietfedrats.
BasicClinPharmacolToxicol2010;106:38995.
13.
LiXH,McGrathKC,NammiS,HeatherAK,RoufogalisBD.
AttenuationofliverproinflammatoryresponsesbyZingiberofficinaleviainhibitionofNFkappaBactivationinhighfatdietfedrats.
BasicClinPharmacolToxicol2012;110:23844.
14.
GaoH,GuanT,LiC,ZuoG,YamaharaJ,WangJ,etal.
TreatmentwithgingeramelioratesfructoseinducedFattyliverandhypertriglyceridemiainrats:Modulationofthehepaticcarbohydrateresponseelementbindingproteinmediatedpathway.
EvidBasedComplementAlternatMed2012;2012:570948.
15.
ChenMH,ChenJC,TsaiCC,WangWC,ChangDC,LinCC,etal.
Shosaikotopreventsliverfibrosisinducedbybileductligationinrats.
AmJChinMed2004;32:195207.
16.
SakaidaI,HironakaK,KimuraT,TeraiS,YamasakiT,OkitaK.
HerbalmedicineShosaikoto(TJ9)increasesexpressionmatrixmetalloproteinases(MMPs)withreducedexpressionoftissueinhibitorofmetalloproteinases(TIMPs)inratstellatecell.
LifeSci2004;74:225163.
17.
KayanoK,SakaidaI,UchidaK,OkitaK.
InhibitoryeffectsoftheherbalmedicineShosaikoto(TJ9)oncellproliferationandprocollagenWorldJTraditChinMed|Volume3|Issue3|JulySeptember201750geneexpressionsinculturedrathepaticstellatecells.
JHepatol1998;29:6429.
18.
ShimizuI.
Shosaikoto:Japaneseherbalmedicineforprotectionagainsthepaticfibrosisandcarcinoma.
JGastroenterolHepatol2000;15Suppl:D8490.
19.
ShimizuI,MaYR,MizobuchiY,LiuF,MiuraT,NakaiY,etal.
EffectsofShosaikoto,aJapaneseherbalmedicine,onhepaticfibrosisinrats.
Hepatology1999;29:14960.
20.
KitadeY,WatanabeS,MasakiT,NishiokaM,NishinoH.
InhibitionofliverfibrosisinLECratsbyacarotenoid,lycopene,oraherbalmedicine,Shosaikoto.
HepatolRes2002;22:196205.
21.
OnoM,MiyamuraM,KyotaniS,SaibaraT,OhnishiS,NishiokaY.
EffectsofShosaikotoextractonliverfibrosisinrelationtothechangesinhydroxyprolineandretinoidlevelsoftheliverinrats.
JPharmPharmacol1999;51:107984.
22.
MiyamuraM,OnoM,KyotaniS,NishiokaY.
EffectsofShosaikotoextractonfibrosisandregenerationoftheliverinrats.
JPharmPharmacol1998;50:97105.
23.
YamashikiM,NishimuraA,NomotoM,SuzukiH,KosakaY.
Herbalmedicine'Shosaikoto'inducestumournecrosisfactoralphaandgranulocytecolonystimulatingfactorinvitroinperipheralbloodmononuclearcellsofpatientswithhepatocellularcarcinoma.
JGastroenterolHepatol1996;11:13742.
24.
ShiotaG,MaetaY,MukoyamaT,YanagidaniA,UdagawaA,OyamaK,etal.
EffectsofShoSaikotoonhepatocarcinogenesisand8hydroxy2'deoxyguanosineformation.
Hepatology2002;35:112533.
25.
LiuXQ,HuXJ,XuHX,ZengXY.
XiaochaihuDecoctionattenuatestheviciouscirclebetweentheoxidativestressandtheALPinactivationthroughLPScatecholaminesinteractionsingut,liverandbrainduringCCI4ethanolinducedmouseHCC.
BMCComplementAlternMed2013;13:375.
26.
YanoH,MizoguchiA,FukudaK,HaramakiM,OgasawaraS,MomosakiS,etal.
TheherbalmedicineshosaikotoinhibitsproliferationofcancercelllinesbyinducingapoptosisandarrestattheG0/G1phase.
CancerRes1994;54:44854.
27.
OkitaK,LiQ,MurakamioT,TakahashiM.
AntigrowtheffectswithcomponentsofShosaikoto(TJ9)onculturedhumanhepatomacells.
EurJCancerPrev1993;2:16975.
28.
LiJ,XieM,GanY.
EffectofXiaochaihudecoctionanddifferentherbalformulationofcomponentoninhibitingH22livercancerinmiceandenhancingimmunefunction.
ZhongguoZhongYaoZaZhi2008;33:103944.
29.
WatanabeS,KitadeY,MasakiT,NishiokaM,SatohK,NishinoH.
EffectsoflycopeneandShosaikotoonhepatocarcinogenesisinaratmodelofspontaneouslivercancer.
NutrCancer2001;39:96101.
30.
TajiriH,KozaiwaK,OzakiY,MikiK,ShimuzuK,OkadaS.
Effectofshosaikoto(xiaochaihutang)onHBeAgclearanceinchildrenwithchronichepatitisBvirusinfectionandwithsustainedliverdisease.
AmJChinMed1991;19:1219.
31.
DengG,KurtzRC,VickersA,LauN,YeungKS,ShiaJ,etal.
AsinglearmphaseIIstudyofaFarEasterntraditionalherbalformulation(Shosaikotoorxiaochaihutang)inchronichepatitisCpatients.
JEthnopharmacol2011;136:837.
32.
SaruwatariJ,NakagawaK,ShindoJ,NachiS,EchizenH,IshizakiT.
TheinvivoeffectsofShosaikoto,atraditionalChineseherbalmedicine,ontwocytochromeP450enzymes(1A2and3A)andxanthineoxidaseinman.
JPharmPharmacol2003;55:15539.
33.
YamashikiM,NishimuraA,HuangXX,NoboriT,SakaguchiS,SuzukiH.
EffectsoftheJapaneseherbalmedicine"Shosaikoto"(TJ9)oninterleukin12productioninpatientswithHCVpositivelivercirrhosis.
DevImmunol1999;7:1722.
34.
OkaH,YamamotoS,KurokiT,HariharaS,MarumoT,KimSR,etal.
ProspectivestudyofchemopreventionofhepatocellularcarcinomawithShosaikoto(TJ9).
Cancer1995;76:7439.
35.
NishimuraN,NaoraK,HiranoH,IwamotoK.
AChinesetraditionalmedicine,shosaikoto(xiaochaihutang),reducesthebioavailabilityoftolbutamideafteroraladministrationinrats.
AmJChinMed1999;27:35563.
36.
NishimuraN,NaoraK,HiranoH,IwamotoK.
Effectsofshosaikoto(xiaochaihutang),aChinesetraditionalmedicine,onthegastricfunctionandabsorptionoftolbutamideinrats.
YakugakuZasshi2001;121:1539.
37.
NishimuraN,NaoraK,HiranoH,IwamotoK.
EffectsofShosaikotoonthepharmacokineticsandpharmacodynamicsoftolbutamideinrats.
JPharmPharmacol1998;50:2316.
38.
LeeJK,KimJH,ShinHK.
TherapeuticeffectsoftheorientalherbalmedicineShosaikotoonlivercirrhosisandcarcinoma.
HepatolRes2011;41:82537.
39.
SatoA,ToyoshimaM,KondoA,OhtaK,SatoH,OhsumiA.
PneumonitisinducedbytheherbalmedicineShosaikotoinJapan.
NihonKyobuShikkanGakkaiZasshi1997;35:3915.
40.
WadaY,KuboM.
AcutelymphoblasticleukemiacomplicatedbytypeChepatitisduringtreatmentandfurtherbyacuteinterstitialpneumoniaduetoShosaikotoin7yearold.
Arerugi1997;46:114855.
41.
KatouK,MoriK.
AutoimmunehepatitiswithdruginducedpneumoniaduetoShosaikoto.
NihonKokyukiGakkaiZasshi1999;37:6416.
42.
TomiokaH,HashimotoK,OhnishiH,FujiyamaR,SakuraiT,TadaK,etal.
AnautopsycaseofinterstitialpneumoniaprobablyinducedbyShosaikoto.
NihonKokyukiGakkaiZasshi1999;37:10138.
43.
DaiboA,YoshidaY,KitazawaS,KosakaY,BandoT,SudoM.
Acaseofpneumonitisandhepaticinjurycausedbyaherbaldrug(shosaikoto).
NihonKyobuShikkanGakkaiZasshi1992;30:15838.
44.
TojimaH,YamazakiT,TokudomeT.
TwocasesofpneumoniacausedbyShosaikoto.
NihonKyobuShikkanGakkaiZasshi1996;34:90410.
45.
NakagawaA,YamaguchiT,TakaoT,AmanoH.
FivecasesofdruginducedpneumonitisduetoShosaikotoorinterferonalphaorboth.
NihonKyobuShikkanGakkaiZasshi1995;33:13616.
46.
MurakamiK,OkajimaK,SakataK,TakatsukiK.
Apossiblemechanismofinterstitialpneumoniaduringinterferontherapywithshosaikoto.
NihonKyobuShikkanGakkaiZasshi1995;33:38994.
47.
OhtakeN,SuzukiR,DaikuharaH,NakaiY,YamamotoM,AmagayaS,etal.
ModulationoflunglocalimmuneresponsesbyoraladministrationofaherbalmedicineShosaikoto.
IntJImmunopharmacol2000;22:41930.
48.
ChenYW,TsaiMY,PanHB,TsengHH,HungYT,ChouCP.
GadoxeticacidenhancedMRIandsonoelastography:NoninvasiveassessmentsofchemopreventionofliverfibrosisinthioacetamideinducedratswithShoSaikoTo.
PLoSOne2014;9:e114756.
- Antifibrogenicm.kan84.net相关文档
- 杆菌m.kan84.net
- Crispm.kan84.net
- Istanbulm.kan84.net
- explicitlym.kan84.net
- Solutionm.kan84.net
- stationm.kan84.net
织梦DEDECMS即将授权收费和维权模式 站长应对的几个方法
这两天在站长群里看到不少有使用DEDECMS织梦程序的朋友比较着急,因为前两天有看到来自DEDECMS,我们熟悉的织梦程序官方发布的公告,将会在10月25日开始全面商业用途的使用DEDECMS内容管理程序的会采用授权收费模式,如果我们有在个人或者企业商业用途的,需要联系且得到授权才可以使用,否则后面会通过维权的方式。对于这个事情,我们可能有些站长经历过,比如字体、图片的版权。以及有一些国内的CMS...

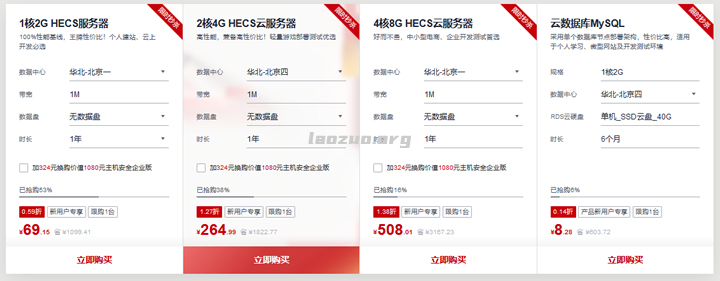
华为云(69元)828促销活动 2G1M云服务器
华为云818上云活动活动截止到8月31日。1、秒杀限时区优惠仅限一单!云服务器秒杀价低至0.59折,每日9点开抢秒杀抢购活动仅限早上9点开始,有限量库存的。2G1M云服务器低至首年69元。2、新用户折扣区优惠仅限一单!购云服务器享3折起加购主机安全及数据库。企业和个人的优惠力度和方案是不同的。比如还有.CN域名首年8元。华为云服务器CPU资源正常没有扣量。3、抽奖活动在8.4-8.31日期间注册并...
青果云(59元/月)香港多线BGP云服务器 1核 1G
青果云香港CN2_GIA主机测评青果云香港多线BGP网络,接入电信CN2 GIA等优质链路,测试IP:45.251.136.1青果网络QG.NET是一家高效多云管理服务商,拥有工信部颁发的全网云计算/CDN/IDC/ISP/IP-VPN等多项资质,是CNNIC/APNIC联盟的成员之一。青果云香港CN2_GIA主机性能分享下面和大家分享下。官方网站:点击进入CPU内存系统盘数据盘宽带ip价格购买地...

m.kan84.net为你推荐
-
同ip站点同IP网站具体是什么意思,能换独立的吗porndao单词prondao的汉语是什么m.kan84.net经常使用http://www.feikan.cc看电影的进来帮我下啊www.kknnn.com求有颜色的网站!要免费的www.hyyan.comdota屠夫怎么玩?从初期到后期的装备是什么?www.45gtv.com登录农行网银首页www.abchina.com,网页源代码网页的HTML代码国风商讯国风网络公司的福利怎么样猴山条约中国近代史领土被割占去了多少,包括战争中失去的和吞并的总数莱姿蔓蕊姿蔓是什么样的牌子来的